Solubility Facts For Kids
Solubility is the ability of a solute to mix with a solvent to form a solution, essential in many chemical and everyday processes.


Do more with AI
Introduction
Solubility is a cool science word that tells us how well one thing can mix into another! 🌊When we add a substance called a solute (like sugar 🍬) to a liquid called a solvent (like water 💧), we can create a solution. For example, if you stir sugar in water, the sugar dissolves and you get sweet water! Different solutes have different solubility levels, so some dissolve better than others. The solubility of a substance can change depending on what liquid you use and the conditions around it. Isn’t that fun? 🚀
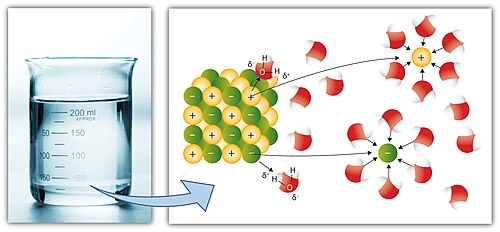
Gallery of Solubility
Types Of Solubility
There are two main types of solubility: solid solubility and gas solubility! 🔍Solid solubility happens when a solid, like salt, dissolves in a liquid. For instance, salt (sodium chloride) dissolves in water very easily. On the other hand, gas solubility occurs when gases, like carbon dioxide, mix with liquids. 🥤When you open a soda can, the fizz is gas that’s dissolved in liquid! Different solutes behave differently, so some solids might dissolve better in warm water while others might not dissolve at all. Cool, right? 🌈
Measuring Solubility
Scientists measure solubility to know how much solute can dissolve in a solvent. 📏They usually express solubility in grams of solute per liter of solvent. For example, if 100 grams of salt can dissolve in 1 liter of water, that’s its solubility! 💧There are special tools like beakers and graduated cylinders to make these measurements more accurate! Scientists sometimes test at different temperatures to see how solubility changes. By doing this, they can discover amazing things about how materials blend and work together! How cool is science? 🤓
Common Solubility Terms
There are some fun terms related to solubility! 💡Here are a few:
- Solute: The substance that dissolves (like sugar or salt!)
- Solvent: The liquid that does the dissolving (like water!)
- Solution: The mixture that forms after dissolving!
- Saturated: When a solvent can't dissolve any more solute!
- Unsaturated: When a solvent can still dissolve more solute!
These terms make it easier to talk about solubility and how things mix together, like a secret science language! 📖
Pressure And Solubility
Pressure is another important factor in solubility, especially for gases! 🌌When you increase pressure, more gas can dissolve in a liquid. Imagine a fizzy drink in a sealed can. The pressure inside keeps the carbon dioxide (the gas) dissolved. When you open the can, the pressure drops, and the gas escapes, creating that bubbly fizz! 🍾Gases, like oxygen, can dissolve in water too, which helps fish breathe! 🐟So, pressure plays a big part in how things mix in our world—just like magic! 🎩
Applications Of Solubility
Understanding solubility is super important! 🏥In medicine, doctors use solubility to create liquid medicines that dissolve the right way. In cooking, bakers need to know how sugar dissolves to make pastries fluffy! 🍰Even in the environment, plants absorb water and nutrients from the soil, which depends on solubility. 🪴Industries use solubility to make drinks, cleaners, and many products we use every day! With this knowledge, we learn how to make and use things better! Wow, science is everywhere! 🌟
Temperature And Solubility
Temperature plays a big role in solubility! 🌡️ Did you know that most solids dissolve better when warm? For example, sugar dissolves faster in hot tea than in iced tea! 🍵However, when it comes to gases, the story changes. Higher temperatures can make gases less soluble! For instance, warm soda loses fizz faster than cold soda. 🥤So, if you want a fizzy drink, keep it cool! It’s like a little science magic happening with heat and mixing! Isn’t it fascinating how temperature affects what dissolves? 🤩
Factors Affecting Solubility
Several factors can affect how well a solute dissolves in a solvent! 🌡️ One big factor is temperature. Warm liquids often dissolve things better than cold liquids. For example, hot tea dissolves sugar faster than ice-cold drinks! Another important factor is the type of solvent we use. Not all liquids mix well with all solutes—like oil and water! 🛢️ Finally, stirring helps mix things better, making the solute dissolve faster! These factors all work together to determine solubility! How interesting! 📚
Solubility In Different Solvents
Not all solvents are the same, and this changes how things dissolve! 🌍Water is called the “universal solvent” because it dissolves many substances, like sugar and salt! 💧But did you know some things don’t mix with water? For example, oil doesn’t mix with water, so you see bubbles instead! 🛢️ Another cool solvent is alcohol, which can dissolve substances that water cannot. Each solvent works differently with different solutes, which is why it’s essential to choose the right one for the task! Let’s experiment and see what happens! 💥
Environmental Impact Of Solubility
Solubility really affects our environment! 🌍When chemicals from factories dissolve in rivers, they can harm plants and animals, like fish. 🐠However, solubility can also help clean things up! For example, some pollutants dissolve in water and can be washed away. Knowing how solubility works helps scientists protect nature! 🌱They study polluted water and can figure out how to clean it up and make it safe again! So, understanding solubility plays an important part in keeping our planet healthy and safe for everyone! 🌎
The Role Of Solubility In Chemical Reactions
Solubility is crucial in chemical reactions! 🔬When substances dissolve in water, they can react with each other to form new substances! For example, when vinegar (an acid) and baking soda (a base) mix, they create bubbles from a gas called carbon dioxide! 🍾This reaction happens best when both substances dissolve in water. Understanding solubility helps scientists create new products and study how reactions occur. Chemistry is like a puzzle, and solubility is one important piece! 🧩