Octet Rule Facts For Kids
The octet rule explains how main-group elements bond to achieve eight electrons in their valence shell, similar to noble gases.

Do more with AI
Introduction
The octet rule is a special rule in chemistry! 🧪It tells us that many elements, especially main-group elements, like to have eight electrons in their outer shell. This makes them stable and happy, just like noble gases such as helium and neon. Imagine these atoms as kids at a party; they want to have enough friends (electrons) to feel great! When they bond together (join up), they share or transfer electrons to reach that magic number of eight! 🌟
Images of Octet Rule
History Of The Octet Rule
The octet rule was first introduced by a scientist named Gilbert Lewis in 1916! 🎉He was trying to explain how atoms combine to form molecules, like building with LEGO bricks. Lewis discovered that atoms become more stable when they have eight electrons in their outer layer, similar to noble gases like argon. This idea helped other scientists understand why certain atoms bond while others do not. Over time, it became an important guideline in chemistry and helped us learn more about how molecules like water (H₂O) and carbon dioxide (CO₂) are formed! 🌍
Significance In Chemistry
The octet rule is super important in chemistry! It helps scientists predict how atoms will interact with each other. When atoms follow this rule, they form strong bonds, creating molecules that make everything from water to sugar! 🍭Understanding the octet rule also helps chemists figure out how to balance chemical equations, which are like recipes for reactions. Knowing this rule helps us to understand reactions in everyday life, like cooking food or cleaning! 🍽️
Exceptions To The Octet Rule
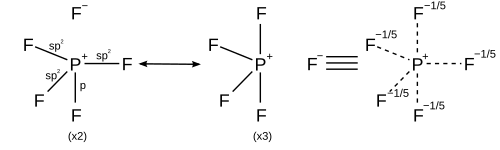
Not all atoms follow the octet rule! 🌈Some elements can be “loners” and be happy with fewer or more than eight electrons. For example, hydrogen and helium only need two electrons to feel stable. Moreover, heavier elements such as phosphorus and sulfur can have more than eight electrons! This is called “expanded octet.” These exceptions are like kids who don't follow the party rules—sometimes they just like to be different! 😜
Impact On Periodic Table Trends
The octet rule also affects the periodic table! 📊Elements are arranged in rows and columns based on their electron configurations! As you move across a period, atoms gain more electrons, aiming for that magical eight! This explains why the reactivity of elements changes; for example, the alkali metals (Group 1) are very reactive because they only have one electron to give away! Meanwhile, the noble gases (Group 18) are non-reactive because they already have eight electrons. Understanding these trends helps us predict element behavior in reactions! 🌟
Applications In Molecular Biology
The octet rule is also important in biology! 🧬It helps scientists understand how DNA and proteins are formed. For example, the atoms in DNA must bond in specific ways to create the double helix structure, which carries genetic information! When atoms follow the octet rule, they create strong bonds between nucleotides in DNA. These strong bonds help life stay stable and grow. Understanding these rules helps researchers develop medicines and treatments, making our world healthier! 💊
Implications For Chemical Bonding
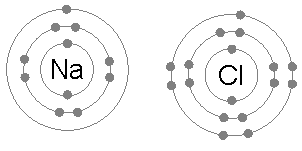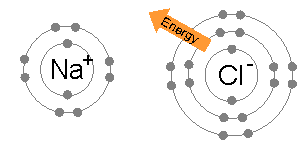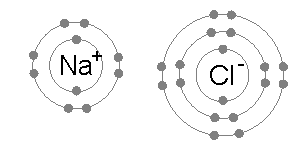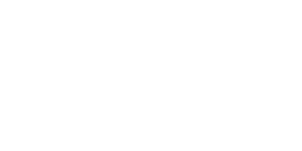
The octet rule impacts how atoms bond together! 🧲When atoms share or transfer electrons to reach eight, they form different types of bonds. If they share electrons, we have a covalent bond (like a friendship bracelet). If one atom takes electrons from another, it creates an ionic bond (a stronger connection). These bonds help form all the materials we see around us—from the air we breathe to the chairs we sit on. So next time you look around, remember that chemical bonding makes it all possible! 🪑
Examples Of Octet Rule In Common Molecules
Let’s explore some common molecules that follow the octet rule! 🌼Water (H₂O) has two hydrogen atoms sharing electrons with one oxygen atom, giving each atom a full outer shell. Another example is carbon dioxide (CO₂), where one carbon atom shares electrons with two oxygen atoms. These molecules follow the octet rule and are essential for life! 🍃Understanding these examples helps highlight how atoms work together to make the world around us! So remember, every time you drink water or breathe, those molecules are playing by the octet rule! 💧
Comparison With Other Electron Configurations
Besides the octet rule, there are other ways atoms can arrange their electrons. 🌀The “duet rule” applies to smaller atoms like hydrogen and helium, which only want two electrons. Then, there’s the “expanded octet” for larger atoms, which can accommodate more than eight electrons! Think of these rules as different party themes; different atoms feel comfortable in different settings. Though the octet rule is the most well-known, knowing about these other configurations can help scientists understand all kinds of chemical interactions! 🎈
Educational Activities To Explore The Octet Rule
Learning about the octet rule can be super fun! 🎨One activity is to create electron dot diagrams! You can draw atoms and their electrons on paper, showing how they bond. Try building models with colorful beads and wires to visualize how atoms connect! 👷♀️ Another great idea is to play “Chemical Bonding Bingo,” where you can match symbols to their corresponding molecules. Engaging in these activities can help you grasp the octet rule and how it works in our everyday lives! Let’s get creative! 🎉
Octet Rule And Valence Shell Electron Pair Repulsion (vsepr)
The octet rule connects to something called VSEPR theory! 🔬VSEPR helps us understand the shapes of molecules based on how electrons are arranged around the atoms. When atoms follow the octet rule, they arrange themselves to keep their electrons apart, just like friends keeping a little space at a dance party. 🕺💃 These shapes influence how molecules react with each other and determine properties like boiling point and melting point. Knowing VSEPR adds depth to our understanding of chemistry and makes learning fun! 🎓
Octet Rule Quiz
Learn more about Octet Rule