Moscovium Facts For Kids
Moscovium is a synthetic element, classified as a heavy, unstable post-transition metal with no practical applications due to its short half-life.

Do more with AI
Introduction
Moscovium is a superheavy element with the symbol Mc and atomic number 115. 🧪It was discovered in 2003 by a team of scientists at the Joint Institute for Nuclear Research in Dubna, Russia, and Lawrence Livermore National Laboratory in California, USA. They named it after Moscow, as "Moscovium" reflects the region's name! This element is special because it doesn't occur naturally and is made in laboratories. Moscovium is part of the group of elements called "post-transition metals" and is known for its rarity. Scientists study moscovium to learn more about how heavy elements behave! 🌟
Gallery of Moscovium
Isotopes Of Moscovium
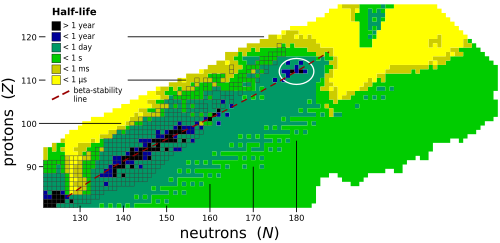
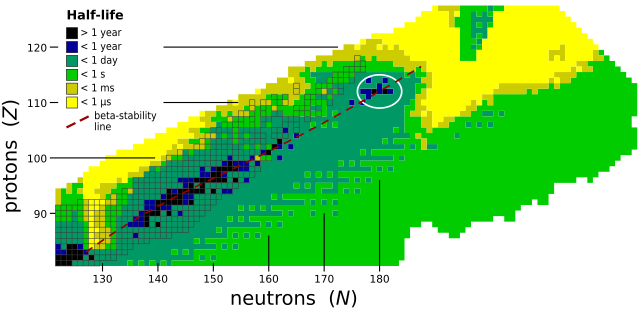
Isotopes are versions of elements with the same number of protons but different numbers of neutrons! Moscovium has only a few isotopes, meaning it can exist in a couple of forms. 🤓The most known isotope is Moscovium-288, but it has a very short life of just about 0.65 milliseconds! This means it quickly turns into other elements, making it very challenging to study. Other isotopes like Moscovium-287 exist, but scientists have mainly focused on those few known versions. Because of its instability, moscovium isotopes help scientists explore nuclear reactions and understand element behavior. 🔬
Discovery Of Moscovium
In 2003, a group of scientists led by Yuri Oganessian discovered moscovium. 🧑🔬 They made it by smashing together atoms of calcium and americium in a particle accelerator. When they collided these atoms, they created moscovium, but only a few tiny amounts! The experiment took place over several hours. Scientists use special techniques to detect it, including watching for its unique particles. They confirmed the discovery after studying the element's radioactive decay. The name "Moscovium" was officially accepted in 2016 by the International Union of Pure and Applied Chemistry (IUPAC). 🏅
Properties Of Moscovium
Moscovium is a really heavy element, with atomic mass around 288 atomic mass units! ⚖️ It is a metal that is believed to be solid at room temperature, although scientists have not studied it extensively due to its rarity. Moscovium is not visible and cannot be found in everyday life because it decays quickly. 🏭It has a very short lifespan, with half-lives lasting less than a second! While researchers expect it to resemble thallium, it's different due to its unique properties and heavy atomic structure. Milliseconds after it's made, it turns into other elements! 🌌
Applications Of Moscovium
Moscovium doesn't have practical applications yet, mainly because it's super rare and only exists for a tiny fraction of a second! 💡Scientists are mainly interested in it for academic research to explore the properties of heavy elements and gain insight into atomic behavior. If researchers find methods to stabilize moscovium, we may discover future uses in materials science or nuclear chemistry! 🏗️ However, majority of studies with moscovium currently focus on its atomic structure and behavior rather than how it might help us in daily life. So, it’s still a big mystery! 🔍
Future Research Directions
Scientists are excited to study moscovium further! 🔬They hope to understand its chemical behaviors better and learn if other isotopes can be created. Some researchers aim to develop new techniques for producing heavier elements with longer lifespans. They want to explore the possibility of stabilizing moscovium or creating compounds! 🧪Future studies will focus on how superheavy elements interact with their environments. As technology advances, lab experiments will help uncover more secrets. Who knows? Moscovium could lead to exciting discoveries about the universe and the building blocks of everything around us! 🌌
Moscovium In Popular Culture
Moscovium is not widely known in pop culture, but it has made some fun appearances! 🎬It has been mentioned in science fiction books and articles exploring the future of elements. Also, its unique name has caught the attention of Internet users keen on learning about elements on the periodic table! 📝Kids interested in science often share interesting facts about moscovium on social media, and some games feature it as an element. As more people learn about it, moscovium might become a fun topic for science trivia and games! Everything about moscovium excites curious minds! 🧠
Chemical Behavior Of Moscovium
Moscovium is expected to behave like other elements in its group, like lead or thallium, but we are not entirely sure yet! ⚗️ In experiments, scientists believe it will have similar chemical traits because it has three electrons in its outer shell. Moscovium might form compounds, but that hasn't been observed directly. The element is predicted to be quite reactive due to its heavy weight and the way its electrons are arranged. Researchers hope to understand those reactions better with more studies! As they study moscovium, we can learn about heavy elements and how they behave differently. 🌈
Safety And Handling Of Moscovium
Moscovium is very unstable, meaning it decays quickly and releases radiation! ⚠️ Because of this, scientists working with it follow strict safety rules to ensure their safety. 🧑🔬 They wear protective gear like gloves and goggles and work in special facilities designed for handling radioactive materials. Because it’s created in tiny amounts, researchers often handle it in small areas. Since moscovium isn't found in nature, there isn't a risk of everyday exposure. Learning about safety helps scientists manage other hazardous materials too! Education about safety while handling unknown elements is essential for conducting exciting research! ⚗️
Comparison With Other Superheavy Elements
Moscovium belongs to a special group called "superheavy elements," which are heavier than lead! 🏋️ These elements are famously known for being unstable and decaying quickly. For example, elements like dubnium and seaborgium are also superheavy! 🌌Scientists compare these elements to understand how they behave, how heavy their nuclei are, and what makes them different. While they share similar properties, each element has unique quirks. Comparing moscovium with its neighbors helps researchers improve their knowledge of the entire periodic table. It's like solving a giant puzzle of nature! 🔍
Moscovium Quiz
Learn more about Moscovium


![Scheme of an apparatus for creation of superheavy elements, based on the Dubna Gas-Filled Recoil Separator set up in the Flerov Laboratory of Nuclear Reactions in JINR. The trajectory within the detector and the beam focusing apparatus changes because of a dipole magnet in the former and quadrupole magnets in the latter.[37]](https://upload.wikimedia.org/wikipedia/commons/thumb/3/34/Apparatus_for_creation_of_superheavy_elements_en.svg/500px-Apparatus_for_creation_of_superheavy_elements_en.svg.png)