Ketone Facts For Kids
Ketones are organic compounds characterized by a carbon atom double-bonded to an oxygen atom, with the general formula R−C−R'.


Do more with AI
Introduction
Ketones are special types of chemical compounds made of carbon, hydrogen, and oxygen! 💧They have a unique structure where a carbon atom is double-bonded to an oxygen atom and single-bonded to two other carbon atoms. This structure makes ketones different from other compounds. A common example of a ketone is acetone, which is found in nail polish remover. 🥰Acetone has the formula C₃H₆O. Ketones are important in chemistry and biology and are found in many everyday products!
Gallery of Ketone
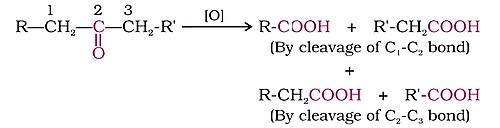
Synthesis Of Ketones
Scientists can create ketones using different methods! One way to make them involves taking alcohols and oxidizing them. 🌱For example, if you start with a three-carbon alcohol and oxidize it, you’ll end up with a three-carbon ketone like propanone! Lab chemists like to experiment with these processes to learn more about how ketones work. Sometimes they even make new types of ketones not seen before! 🔬
Ketones In Metabolism
When we eat food, our body breaks it down into energy. 🍽️ One of the ways our body processes fats for energy is by creating ketones! This is especially important during special diets like the ketogenic diet. 🚴In this diet, people eat fewer carbs and more fats, which encourages the body to produce more ketones. This way, ketones become a key energy source! Our bodies are amazing, aren't they? 🌟
Nomenclature Of Ketones
Nomenclature is a fancy word for naming things! 📝Ketones are named based on the number of carbon atoms in their longest chain. For example, if there are three carbon atoms, it’s called propanone (that’s acetone)! If there are four carbon atoms, it’s called butanone. This is important for chemists when talking about different ketones. They follow a set of rules, so each ketone has a unique name. This helps everyone understand which ketone they’re talking about! 🌈
Biological Role Of Ketones
Did you know that ketones play an important role in our bodies? 🔍When we don’t eat enough carbs, our bodies switch to burning fat for energy. This produces ketones! 🥑These are called "ketone bodies" and can be used by our brains and muscles for energy, especially during exercise or fasting. This process is known as ketosis. Understanding how our bodies use ketones is important for keeping us healthy! 🏋️♀️
Reactions Involving Ketones
Ketones like to react with other chemicals! 🔄They can participate in many amazing chemical reactions. One important reaction is when ketones are produced through oxidation, which means adding oxygen. They also react with alcohols to form compounds called hemiacetals! 🥳In cooking, ketones are involved in baking. When cookies are baked, the heat can change some sugars into ketones, giving treats their yumminess! 🍪
Chemical Structure Of Ketones
The chemical structure of a ketone looks like this: R−C(=O)−R'. The “R” and “R’” represent groups of carbon atoms, while the “C” is the carbon atom in the center. The “O” stands for oxygen. 🌍This structure means that ketones have a carbon double-bonded to oxygen (C=O), which is called a carbonyl group. This carbonyl group is what makes ketones special! 📐So, if you see a structure like this in chemistry, you know it's a ketone!
Physical Properties Of Ketones
Ketones have some interesting physical properties! 🧊Most ketones are colorless liquids at room temperature. They usually have a sweet smell, like many fruits! 🍉Ketones can mix well with water, which is important for how they behave in our bodies and the environment. Acetone, for example, has a boiling point of 56 degrees Celsius (132.8 degrees Fahrenheit), which is pretty low! This means it can easily turn from a liquid into a gas. 🌀
Safety And Handling Of Ketones
While ketones are useful, they can be dangerous in certain situations. ⚠️ Acetone, for instance, can be flammable, which means it can catch fire easily! 🔥It’s important to handle ketones carefully and use them in well-ventilated areas to avoid inhaling fumes. Always wear gloves and goggles when working with them. Safety first! 🥽💖
Applications Of Ketones In Industry
Ketones are super useful in many industries! 🛠️ They are commonly used as solvents, which help to dissolve other substances. For example, acetone is often used in nail polish removers and paint thinners. 🌈Ketones are also found in the production of plastics, fragrances, and even pharmaceuticals! Every time you see products with these characteristics, they may contain ketones, which help make our lives easier! ⚙️
Ketones Vs. Other Organic Compounds
Ketones are one type of organic compound, which means they contain carbon! 🌳But they're not alone; there's a whole family of organic compounds, like alcohols and aldehydes! Alcohols contain an -OH group, while aldehydes have the carbonyl group at the end. The coolest part is seeing how these compounds react differently! 🧪Each has its own unique characteristics. Ketones are special with their structure but share similarities with other organic compounds, like aromas and chemical properties.