Formic Acid Facts For Kids
Formic acid, also known as methanoic acid, is the simplest carboxylic acid with the formula HCOOH, found in nature and produced by ants, and has various industrial uses.


Do more with AI
Introduction
Formic acid is a special kind of acid, and it’s the simplest one! 🌱Its chemical formula is HCOOH, which means it has one carbon atom (C), two oxygen atoms (O), and two hydrogen atoms (H). Formic acid is found in nature, especially in ant bites! 🐜When ants feel threatened, they squirt formic acid, which gives a little sting. It’s named "formic" because it comes from the Latin word for ant, "formica." People also use formic acid in the industry for different things! Let’s explore more about this fascinating little acid! 🔍
Gallery of Formic Acid
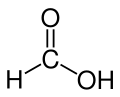
Chemical Structure
The chemical structure of formic acid is pretty simple! 🧪Imagine a line connecting atoms: H–C(=O)–OH. Here, "C" is the center of our structure, with one hydrogen (H) atom on one side and a "carboxyl group" (–COOH) on the other! The "O" in the carboxyl group has a double bond (C=O), which means it’s connected tightly to the carbon atom. This structure allows formic acid to act like an acid! When dissolved in water, it can donate a hydrogen ion (H+). This is what makes substances acidic! 🌊
Production Methods
Formic acid can be made in a few different ways! 🔄One common method is by using carbon monoxide and sodium hydroxide together in a special reaction. Another way is through the fermentation of sugars, which means letting tiny living things, called bacteria, eat sugar and produce formic acid as a byproduct. 🍍Farmers often use this method! Additionally, it can be found in the urine of many insects. So, next time you see an ant, think about the amazing formic acid it might carry with it! 🐜
Chemical Properties
Formic acid can react with many substances! 🔥It's known to be a weak acid, meaning it doesn't release many hydrogen ions. But, it can still participate in some cool chemical reactions, like making esters when mixed with alcohol! 🥃It can also react with bases like sodium hydroxide (NaOH) to form a salt called sodium formate. Additionally, it can break down with heat into carbon monoxide (CO) and water (H2O), which means it can be part of important chemical processes in labs. Isn’t chemistry interesting? 🧬
Physical Properties
Formic acid is a clear, colorless liquid that has a strong, pungent smell, kind of like vinegar! 🤢It has a boiling point of 100.8 °C (213.4 °F), which means it can turn into gas when heated. Formic acid is also soluble in water, which means it mixes well with it. 💧The taste is very sour, like a super sour candy! 😝The melting point is around 8.4 °C (47.1 °F), so it can turn into a solid at really cold temperatures. Remember, even though it’s found in nature, it can be dangerous, so we must be careful! ⚠️
Safety And Handling
Safety is super important when dealing with formic acid! 🛡️ Always wear gloves and goggles if you’re in a lab using it! If it spills, it can hurt your skin, so be careful! 💔If someone gets some on their skin, they should wash it off with lots of water right away. If it gets in your eyes, rinse with water and tell an adult! Always follow the instructions on how to use it safely. Remember, respect chemicals and they’ll respect you! ✅
Environmental Impact
Formic acid has both positive and negative effects on the environment. 🌍It can help in the decomposition of plant matter, returning nutrients to the soil, which is helpful for plants to grow! 🌱However, too much formic acid in the environment, especially from pollution, can harm plants and animals. Scientists are studying ways to use formic acid safely so that it benefits ecosystems instead of harming them. They strive to keep our planet healthy for everyone! 🌈
Uses And Applications
Formic acid is super useful! 🎉It’s mostly used in agriculture to preserve animal feeds, keeping them fresh and nutritious. 🐄People also use formic acid in the textile industry to dye fabrics and in the leather industry for tanning hides. Even the food industry uses it to give some foods a tangy taste! 🍋Additionally, it plays a role in cleaning agents and can help humans in laboratories for various experiments. Formic acid helps keep the world running smoothly in many ways! 🌍
Historical Significance
Formic acid has a surprising history! 📜It was discovered in the 1600s by a scientist named John Ray, who found it in ant bodies. Later, in the 18th century, a chemist named Antoine Lavoisier studied it more. He helped make chemistry a scientific field! Today, formic acid is important for industry and research, showing us how curious humans can discover amazing new things from even the tiniest insects! 🐜Thanks to these discoveries, we understand the world around us better! 🌟
Biological Role And Toxicity
In nature, formic acid helps certain animals! 🐜Ants use it to protect their colonies from enemies and as a way to communicate with each other. 🗣️ However, it can be toxic to humans and animals in high amounts. If someone touches or drinks a lot of formic acid, it can cause burns and other problems. Despite some risks, scientists study it to find ways to use it safely for medicines and other good things! Always ask an adult if you find some formic acid, just to be safe! 🚨
Formic Acid Quiz
Learn more about Formic Acid