Chlorofluorocarbon Facts For Kids
Chlorofluorocarbons (CFCs) are chemicals made of carbon, hydrogen, chlorine, and fluorine that were once commonly used in refrigeration and aerosol products but are harmful to the ozone layer, contributing to increased UV radiation exposure.
Do more with AI
Introduction
Chlorofluorocarbons (CFCs) are special chemicals made of carbon, chlorine, fluorine, and hydrogen. 🌍They were first created in the 1920s and were used in many products like fridges, air conditioners, and spray cans. CFCs are amazing because they can keep things cool! ❄️ However, scientists found out that they harm the Earth's ozone layer, which protects us from the sun's harmful rays. ☀️ Today, many countries are trying to reduce their use to help our planet. Learning about CFCs helps us understand how to be kind to our environment! 🍃
Gallery of Chlorofluorocarbon
Production And Decomposition
Making CFCs used to be a common practice in factories! 🚧The process involved mixing chemicals in a special way. However, because they are bad for our environment, many factories stopped producing them. ☠️ Now, scientists are studying how CFCs break down in the atmosphere. When CFCs reach the stratosphere, they break apart due to sunlight! ☀️ This process releases chlorine atoms, which can destroy ozone molecules. This means CFCs can remain in the environment for many years—up to 100 years! 🌱That’s why cutting how much we use is essential for our Earth’s health! 🌍
Historical Development And Uses
CFCs were invented in the 1920s by a scientist named Thomas Midgley, Jr. 🔬At first, they were seen as the perfect solution for refrigeration and air conditioning. CFCs made cool air possible without being dangerous like other chemicals used before. 🛠️ By the 1970s, CFCs became super popular in products like spray bottles and foam insulation! However, in the 1980s, scientists discovered that these chemicals were causing problems for the ozone layer. 🌍Since then, many countries have worked hard to stop using CFCs. This shows how science helps us understand and improve our world! 🌞
Current Research And Future Trends
Today, researchers are studying ways to make the environment even better! 🌿They are looking for new, safer chemicals to use instead of CFCs. This research is important because inventions can help cool things down without harming the ozone layer! 🌍Scientists are also working on ways to recycle old refrigerants so we can use them again, reducing waste. ♻️ The future for our planet looks brighter as we learn and create solutions that protect both our health and the environment. Let’s all do our part to keep our Earth healthy for years to come! ✨
Alternatives To Chlorofluorocarbons
So, if CFCs are bad for the environment, what do we use instead? 🤔Scientists found some great alternatives like Hydrofluorocarbons (HFCs) and other safer chemicals! 🧊These alternatives can cool things down without destroying the ozone layer. For example, HFC-134a is used in cars and refrigerators. 🚗☃️ Some companies are also using natural gases like propane, which are better for our planet. 🍃It’s great to see how inventiveness can help us take care of the Earth. Always remember that using safer options is a big win for both us and the environment! 🌍
Chlorofluorocarbons In The Atmosphere
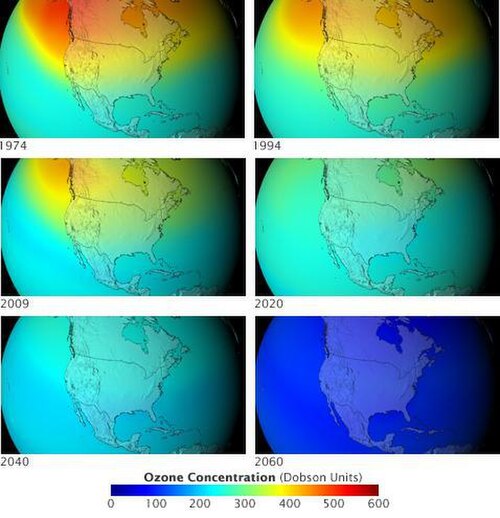
Once CFCs are released into the atmosphere, they can travel very high up! 🌬️ It can take them years, but they eventually reach the stratosphere, where they become a problem for the ozone layer. ⚠️ For every molecule of CFC that breaks apart, it can destroy thousands of ozone molecules! This is why CFCs are considered 'ozone-depleting substances.' 🏭 Scientists monitor the levels of CFCs in the atmosphere using special devices. 📈Thanks to global efforts to reduce their use, the amount of CFCs is dropping, helping to protect the ozone layer for all of us! 🌼
Regulation And International Agreements
To protect our atmosphere, countries created important rules called treaties! One famous treaty is the Montreal Protocol, signed in 1987. 📝This agreement aimed to phase out the use of harmful substances like CFCs. More than 190 countries agreed to stop using most CFCs. 🌏The global teamwork helped reduce the number of CFCs in the atmosphere, showing how people can work together to solve big problems! 🤝Even though it’s been years since CFCs were banned in many places, scientists are still keeping an eye on the recovery of the ozone layer. 📊
Health Effects And Safety Considerations
CFCs can also affect our health! 😮When CFCs leak, they can harm the air we breathe, leading to potential breathing difficulties or skin problems due to increased UV rays. 🥵But good news! Since many of these harmful substances were reduced, health risks have improved. Then there’s the importance of safety when using chemicals! 🧤Always follow safety instructions and remember that even things that seem fun, like sprays, can be harmful if not used properly! 🚫Learning about safe practices helps keep everyone healthy and happy! 🎉
Chemical Structure Of Chlorofluorocarbons
CFCs have a unique chemical structure that gives them their cool properties! 🧪The basic formula for CFCs is CFCl3 or something similar, which means they can have different combinations of carbon (C), chlorine (Cl), and fluorine (F). For example, Freon-12 (CCl2F2) is one type of CFC. The more chlorine and fluorine atoms a CFC has, the stronger it is at cooling things down. ❄️ When you look at it closely with advanced tools, you see a molecule shaped like a little puzzle! 🧩This structure helps them float up into the atmosphere, where they can cause problems for the ozone layer. ⚠️
Environmental Impact Of Chlorofluorocarbons
CFCs may be great for keeping things cool, but they have a big problem! When these gases reach the atmosphere, they can break down the ozone layer. 🌌The ozone layer is like a safety shield around our Earth that protects us from the sun's harmful ultraviolet (UV) rays. 🌞Too much UV exposure can cause skin cancer and other problems for both humans and animals. 🐢Fortunately, thanks to the efforts of scientists and governments around the world, the ozone layer is slowly healing! 🌿By using less harmful chemicals, we can protect our planet for future generations. 🌍


![The warming influence of greenhouse gases in the atmosphere has increased substantially in recent years. The rising presence of carbon dioxide from fossil fuel burning is the largest overall driver. The relatively smaller but significant warming impact from releases of the most abundantly produced CFCs (CFC11 and CFC12) will continue to persist for many further decades into the future.[12][13]](https://upload.wikimedia.org/wikipedia/commons/thumb/c/c4/1979-_Radiative_forcing_-_climate_change_-_global_warming_-_EPA_NOAA.svg/500px-1979-_Radiative_forcing_-_climate_change_-_global_warming_-_EPA_NOAA.svg.png)