Chemical Kinetics Facts For Kids
Chemical kinetics studies the rates of chemical reactions and the factors affecting those rates, helping us understand and control how substances change.


Do more with AI
Introduction
Chemical kinetics is a fun part of science that studies how fast chemical reactions happen! 🧪🔍 It helps scientists understand why some things react quickly, while others take their time. The speed of reactions can change based on different factors, like what materials are involved and their surroundings. Imagine baking a cake! 🎂The ingredients mix quickly when stirred, but the cake takes time to bake in the oven. By studying chemical kinetics, we learn how to speed things up or slow them down, making it an important topic in chemistry! 🌟
Gallery of Chemical Kinetics
Collision Theory
Collision theory explains that for a reaction to happen, particles must collide with each other! 🤝But not just any bump will do! They need to collide with enough energy, like a strong high-five! 👋If they don't, they just bounce off each other! For instance, think of marbles rolling together. If they don’t hit each other hard enough, nothing happens. When they do, though, sparks fly! ⚡Understanding this helps scientists create the best conditions for reactions to happen, making all sorts of things from food to fuels! 🍳⛽
Transition State Theory
In the world of chemistry, every reaction goes through a transition state! It's like a beautiful butterfly emerging from a cocoon! 🦋When particles collide, they transform into something new. The transition state is this tricky moment when they’re not quite one thing or the other—it’s super special and has high energy! ⚡If particles have enough energy when they collide, they can reach this state and then change into the final products! This theory helps chemists understand and control how reactions happen at the tiniest levels. 🧬
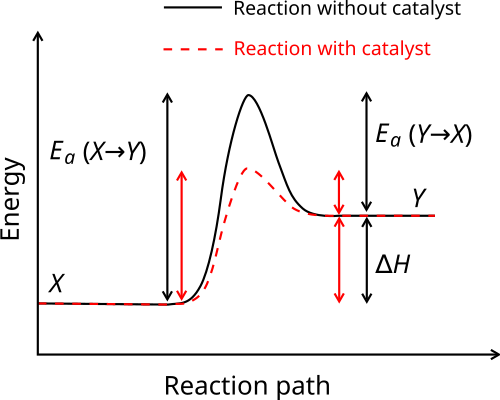
Catalysis And Inhibition
Catalysts are like cheerleaders for chemical reactions! 🎉They help speed things up without being used up themselves! For example, enzymes are special catalysts found in our bodies that speed up digestion! 🍽️ On the other hand, inhibitors are the opposite; they slow down reactions. Picture a stop sign blocking traffic! 🛑Some medicines work as inhibitors, making processes slower to help us heal. By studying both catalysts and inhibitors, scientists can control chemical reactions better, making them safer and more efficient! 🧪⚗️
Temperature And Reaction Rates
Temperature plays a big role in how fast reactions go! 🌡️ When it's warm, particles move around quickly, dancing and bumping into each other more often! 💃This means they are more likely to react. For example, stirring sugar into hot water makes it dissolve faster than in cold water! 🥤In fact, many reactions double their speed for every 10 degrees Celsius increase. That's cool, right? 😎Scientists use this knowledge to do everything from baking bread 🍞 to creating energy in chemical plants! 🔋
Experimental Methods In Kinetics
Scientists use special methods to study chemical kinetics! 🔍One common way is to measure how much of a substance disappears over time. They might use color change, bubbles formed, or differences in weight! For example, in a reaction producing gas, they can capture the gas in a balloon 🤡 and measure its size! They also use high-tech tools like spectrophotometers that measure light passing through solutions. 💡By using these methods, researchers learn all about how fast reactions are happening, helping make new discoveries in science every day! 🌈
Factors Affecting Reaction Rates
Several things can change how fast a reaction goes! ⚡First, the temperature matters. Hotter temperatures usually speed things up, while cooler ones slow them down. 🌞❄️ Next, the concentration of materials matters too. More baking soda in vinegar creates a faster fizz! 🍶The surface area counts as well; powder mixes faster than chunks. Lastly, adding catalysts can hurry things along! 🛠️ Catalysts are special substances that make reactions faster without getting used up. Understanding these factors helps scientists control reactions better for many uses!
Applications Of Chemical Kinetics
Chemical kinetics has exciting real-world applications! 🛠️ It helps create everyday products like medicines, cleaning supplies, and fuels! For instance, researchers in laboratories study reaction rates to make sure cold medicines work effectively when you need them! 🦠Kinetics is especially important in industries too! In food production, knowing how fast things spoil can lead to better preservation methods! 🎂Even environmental scientists use kinetics to understand pollution rates and find solutions to protect our planet! 🌍It’s amazing how this branch of chemistry makes our lives better every day! 😊
Fundamentals Of Chemical Kinetics
In chemical kinetics, we focus on the rate of a reaction, which means how fast or slow it happens. 🌡️ The rate is measured by how much of a substance is used up or produced in a specific time! For example, if you mix vinegar and baking soda, they fizz and create bubbles! 🧼The fizz makes carbon dioxide gas, and we can see how fast it happens by timing the bubbling. It's important to know these rates to understand reactions better and to help in things like making medicines or cleaning products! 💊✨
Rate Laws And Reaction Mechanisms
Rate laws are like secret formulas that help us understand how the speed of a reaction depends on the materials involved. 📊For example, if we have a reaction between A and B, the rate law tells us if A or B is making it faster! 🔑Sometimes, reactions happen in steps, called reaction mechanisms. Each step can be faster or slower than the others! Imagine a race where some runners stop to tie their shoes! 🏃♂️🏃♀️ Learning about these processes helps scientists know how to control reactions for things like making new products or medicines! 💊
Real-world Examples Of Kinetic Studies
There are many fun examples of chemical kinetics in action! 🌟One famous example is the reaction between hydrogen and oxygen to make water! 💧Scientists studied its speed to create engines that power rockets! 🚀Another example is cooking—when baking bread, knowing how temperature affects yeast helps make the fluffiest loaves! 🍞Lastly, scientists worked on the just-right timing of fireworks to create the most beautiful displays! 🎆By studying these phenomena, researchers have created exciting advancements in technology, health, and much more! Chemical kinetics truly helps us explore the world! 🌏✨
Chemical Kinetics Quiz
Learn more about Chemical Kinetics