Ammonium Chloride Facts For Kids
Ammonium chloride (NH4Cl) is an inorganic chemical compound that is white, crystalline, and mainly used in fertilizers and food products.

Do more with AI
Introduction
Ammonium chloride (NH₄Cl) is a cool chemical! 💧It's a white powder that can easily dissolve in water. You might find it in many places, like in food and even in some medicines. This compound comes from combining ammonia (NH₃) and hydrochloric acid (HCl). Ammonium chloride can be found in countries like China and India where it's made in big factories. It’s used to create many important items. But, remember, while it's very useful, it should always be handled carefully! 🌍
Gallery of Ammonium Chloride
Analytical Methods
Scientists have special ways to measure ammonium chloride! 🧪One method involves using a tool called a spectrophotometer, which helps them see how the chemical interacts with light. Another way is to use titration, where they carefully add other substances to find out how much ammonium chloride is present. These methods help researchers make sure products are safe and of good quality! 📊It’s all about finding the right balance.
Historical Context
Ammonium chloride has been known for a long time! 🌟The ancient Egyptians used it as a fertilizer. Scientists started studying it in detail in the 18th century, as they began discovering and classifying all kinds of chemicals! One famous chemist, Antoine Lavoisier, helped improve our understanding of elements! Today, it’s still an important compound in labs and classrooms around the world. 🏫
Environmental Impact
Ammonium chloride is usually safe for the environment! 🌳It helps plants grow when used in fertilizers. However, if too much is used, it can run off into rivers and lakes, which might hurt fish. 🐟That’s why farmers need to use it wisely! It doesn’t mostly pollute the air, so that's good too! Always remember, balance is key when we use anything in nature! 🌍
Uses And Applications
Ammonium chloride has many fun uses! 😄You can find it in food, especially in snacks like salt and vinegar chips. It’s also used in fertilizers to help plants grow better. 🌱Some medicines contain ammonium chloride to help kids with coughs! When a person has a cold, it can help them breathe easier. It's even used in fireworks to make beautiful colors in the sky! 🎆Wow!
Synthesis And Production
To make ammonium chloride, scientists mix ammonia gas with hydrochloric acid! It looks like mixing different colors of paint. 🎨This happens in big factories where it's produced from natural sources. 🌱The process is safe with the right precautions. Most of the world’s ammonium chloride is produced in countries like China, India, and the United States. Once it’s made, it can be packaged and sent to stores or used in many products! 📦
Market And Economic Aspects
The ammonium chloride market is quite big! 💰It's used in many industries, from agriculture to medicine. China and India are the largest producers, exporting it worldwide. Citizens around the globe use ammonium chloride in everyday products, helping boost economies! 🌎As more people learn about its benefits, the demand for ammonium chloride continues to grow. It’s a small powder with a big impact! 🌟
Reactions And Chemical Behavior
When ammonium chloride is heated, it can break down into ammonia and hydrochloric acid. It's like a magic trick! 🎩This reaction is used to create cool displays in chemistry lessons! It also can react with sodium hydroxide, producing ammonia gas. Scientists study these behaviors to understand how different chemicals react! Ammonium chloride can also create a cooling effect when dissolved in water - that’s why it's often found in instant cold packs! ❄️
Health And Safety Considerations
While ammonium chloride is useful, it’s important to be safe! Always wash your hands after touching it. 👋Kids should ask an adult to help when using anything that involves ammonium chloride! If someone eats too much, it can cause tummy aches. 🤢It’s best to keep it away from babies and pets! Wearing gloves and goggles is a good idea for anyone working with it. Safety first! 🚷
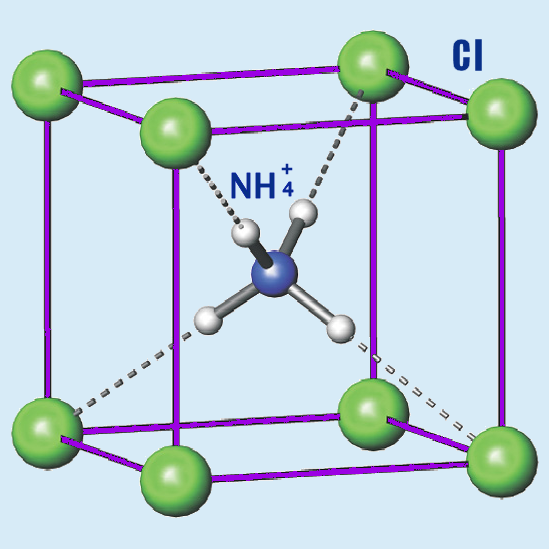
Chemical Structure And Properties
The chemical formula for ammonium chloride is NH₄Cl. This means it has one nitrogen atom (N), four hydrogen atoms (H), and one chlorine atom (Cl). Nitrogen is a gas, while chlorine is a greenish gas that can be very dangerous! 🏭However, when they form ammonium chloride, they become a safe white powder! It dissolves really well in water, creating a yummy salty taste! ☠️ It also has a property called "hygroscopic," meaning it can attract moisture from the air.
Derivatives And Related Compounds
Ammonium chloride has some cool relatives! One of them is ammonium sulfate (NH₄)₂SO₄, which is also used in fertilizers! 🌾Another cousin is ammonium bicarbonate (NH₄HCO₃), great for baking! 🍞These related compounds share some similar properties, but they are also unique in their uses and how they behave in different situations! Chemistry is like a family tree, full of surprises! 🌳


