Nitrate Ion Facts For Kids
Nitrate is a polyatomic ion with the chemical formula NO₃⁻, consisting of one nitrogen atom and three oxygen atoms, and plays a vital role in plant nutrition and the nitrogen cycle.

Do more with AI
Introduction
Nitrate is a special kind of ion made of nitrogen and oxygen! 🧪It has the chemical formula NO₃⁻, meaning one nitrogen atom (N) and three oxygen atoms (O). Nitrate is a negative ion, which means it has more electrons than protons. You can find nitrate in soil, water, and even in our bodies! It is important for plants because they use it to grow big and strong. 🌱Nitrate can also be found in many foods we eat, like spinach and beets. Learning about nitrate helps us understand how nature works! 🌍
Images of Nitrate Ion
Nitrate In Explosives
Did you know nitrate is used in explosives? 💥That's right! Certain types of nitrates, like ammonium nitrate, can create powerful explosions when mixed with other materials. This is useful in mining, construction, and even in making fireworks! 🎇However, it's important that only trained professionals handle these materials, as they can be very dangerous. The amazing part is that the same nitrate that helps plants grow can also create big bangs when used in the right way! ⚡
Sources Of Nitrate Ions
Nitrate ions come from various sources. 🌿One major source is fertilizers used in farming. Farmers spread these fertilizers on their fields to help crops grow better! Nitrate can also be found in animal waste, decomposing plants, and even in some natural water sources like rivers and lakes. 🌊When it rains, nitrate can wash into soil and water systems. Some foods, like leafy greens and cured meats, are also rich in nitrate. So next time you eat vegetables, you’re getting some nitrate ions! 🥗
Nitrate In Food And Health
Nitrate can be found in many of the delicious foods we eat! 🥦Vegetables like spinach, lettuce, and beets are particularly rich in nitrate. When we eat these foods, our bodies convert some of the nitrate into nitrite, which helps our blood transport oxygen. But we need to be careful, as too much nitrite can be harmful. 🩸Most people get the right amount of nitrate from a balanced diet. Eating a variety of colorful vegetables ensures we stay healthy while enjoying tasty meals! 🍽️
Role Of Nitrate In Fertilizers
Nitrate plays a big role in fertilizers! 🌾Fertilizers are substances added to soil to help plants grow healthy and strong. Nitrates provide essential nutrients that plants need to make their own food through a process called photosynthesis. This process helps them produce energy and grow tall! 🌱Many farmers use nitrate-based fertilizers to improve the yield of their crops. Plants like corn and wheat especially love nitrates. When used properly, fertilizers help feed the world by providing enough fruits and vegetables for everyone! 🍅
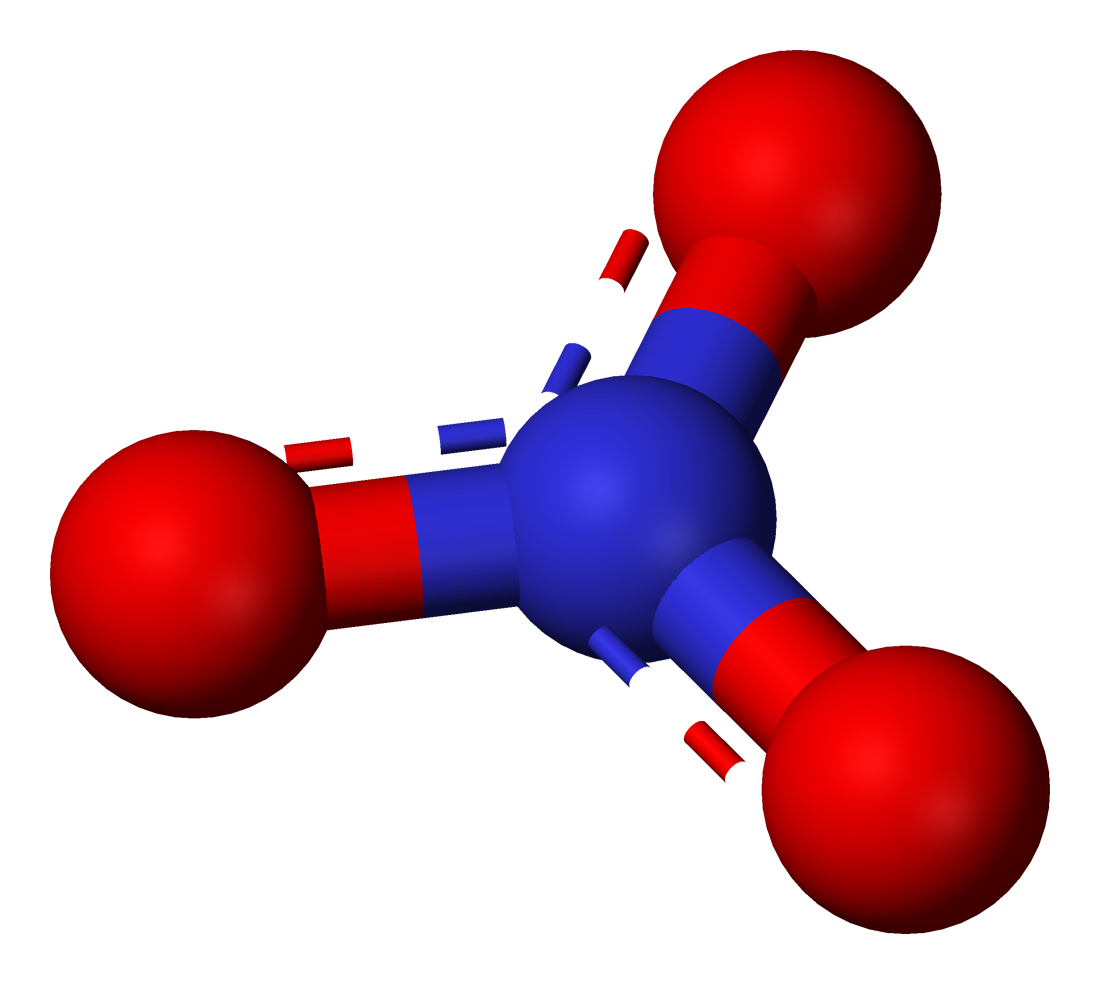
Chemical Structure Of Nitrate Ion
Nitrate has a cool shape! Its chemical structure consists of one nitrogen atom surrounded by three oxygen atoms. ⚛️ The nitrogen atom is in the center, while the oxygen atoms are at the corners, creating a triangular pattern known as a trigonal planar structure. This shape helps nitrate ion react easily with other substances. Nitrate also carries a negative charge because it has gained an extra electron. This negative charge allows it to bond with other positively charged ions, which is very important in biology and chemistry! 🔬
Regulation And Safety Of Nitrates
Nitrates must be handled safely, just like any other chemicals! 🌈Governments and organizations set rules to make sure people use nitrates properly, especially in fertilizers and food production. They check how much nitrate is allowed in drinking water to keep everyone healthy. Too much nitrate can be harmful, so these regulations help protect people and the environment. 🛡️ Following these safety guidelines ensures that we can enjoy the benefits of nitrates while keeping ourselves and nature safe! 🌎
Historical Use Of Nitrate Compounds
Nitrate compounds have been used for a long time in human history! 🕰️ Archaeologists discovered that ancient civilizations used saltpeter (potassium nitrate) to make gunpowder, which helped in wars and hunting. People also used nitrates for preserving food, especially meats, to keep them fresh and flavorful. 🍖Even in the Middle Ages, people recognized nitrates' importance! Today, we continue to find different uses for nitrates in agriculture, medicine, and more. Learning about the past helps us appreciate how nitrates affect our daily lives! 🚀
Nitrate Vs. Nitrite: Key Differences
Nitrate and nitrite are often confused because they are related! 🧪Nitrate (NO₃⁻) has one nitrogen atom and three oxygen atoms, while nitrite (NO₂⁻) has one nitrogen atom and two oxygen atoms. This small difference makes a big impact! Nitrate can be found in foods like leafy greens, while nitrite is often used in processed meats like bacon. 🍔Nitrite can be harmful in large amounts, so we should eat it in moderation. Both have essential roles in our bodies, but remembering these differences helps us make good food choices! 🥗
Nitrate Reduction In Biological Systems
Living organisms, like plants and animals, can reduce nitrate in their bodies! 🌱This means they can change nitrate into different forms, like proteins. Plants use nitrate from the soil, and through a process called "nitrate reduction," they convert it into a substance called amino acids, which are building blocks for proteins! 🧬Animals also take in these amino acids when they eat plants or other animals. This helps everyone grow and stay healthy! Isn't nature amazing? 🌳
Environmental Impact Of Nitrate Pollution
Nitrate pollution can be a big problem for the environment! 🌍When too much nitrate enters rivers and lakes, it can cause something called "algal blooms." These blooms happen when algae grow too quickly, covering the water's surface and blocking sunlight for other plants. 🌊This makes it hard for fish and other creatures to survive. Too much nitrate can come from fertilizers washing into water bodies. That's why we must use fertilizers carefully to protect our environment and keep our waters clean and healthy! 🐟
Nitrate Ion Quiz
Learn more about Nitrate Ion




![Excessive nitrate and phosphate concentrations measured in the Pacific Ocean[citation needed]](https://upload.wikimedia.org/wikipedia/commons/thumb/1/19/Nitrate_and_Phosphate_Pacific_Ocean.jpg/500px-Nitrate_and_Phosphate_Pacific_Ocean.jpg)