Electron Shell Facts For Kids
An electron shell is a layered space around an atom's nucleus where electrons are likely to be found, influencing the atom's behavior and interactions.

Do more with AI
Introduction
Electrons are tiny particles that spin around the center of an atom, called the nucleus! 🌌The nucleus is made of protons and neutrons, and the electrons live in special places called electron shells. Imagine these shells like the rings of a racetrack, where electrons zoom around at different distances from the nucleus. There can be many shells, but the inner ones are filled first. Understanding these shells helps scientists learn about how atoms behave and how they combine to form different substances. ⚛️ So, next time you hear about atoms, remember the little electrons racing around their shells!
Images of Electron Shell
Electron Configuration
Electron configuration is like a recipe that tells us how electrons are arranged in their shells! 📜For example, the configuration for hydrogen (H), which has one electron, is 1s¹, meaning it has 1 electron in the first shell! For oxygen (O), the configuration is 1s² 2s² 2p⁴; this shows that it has 2 electrons in the first shell and 6 in the second! 🍏Understanding electron configuration helps scientists predict how elements will bond and create new materials!
Principal Quantum Numbers
Every electron shell has a special number called the principal quantum number, or simply "n." 🎨 This number tells us where the shell is located and how much energy it has. For the first shell (closest to the nucleus), n = 1. The second shell has n = 2, and it continues increasing. 🌈The higher the number, the farther the shell is from the nucleus and the more energy it has! So, electron shells close to the nucleus are like cozy, low-energy homes, while those farther away are like high-energy towers!
Structure Of Electron Shells
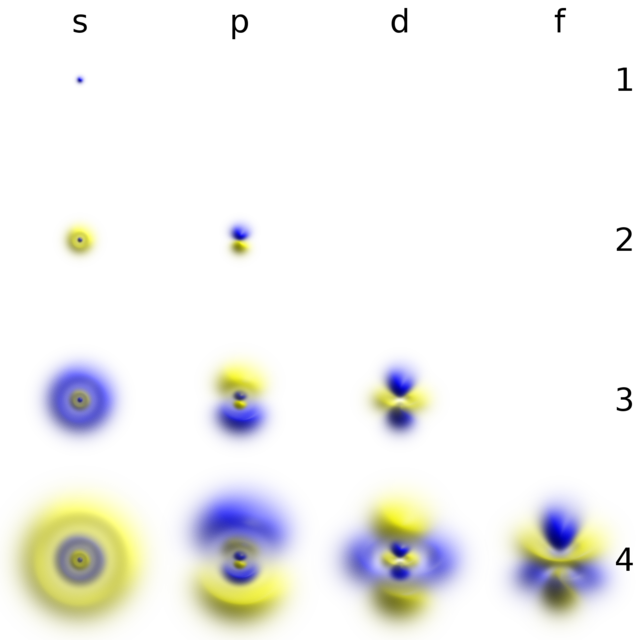
Think of electron shells as layers of an onion! 🧅Each layer has a different distance from the nucleus. The first shell can hold 2 electrons, the second can hold 8, the third can hold 18, and this continues on. There are also different types of sub-shells within the shells that have shapes: spherical (s), dumbbell-shaped (p), and more! 🤹These shapes help describe how electrons are arranged, making it fascinating to learn about how electrons behave in each shell!
Definition Of Electron Shells
Electron shells are like cozy homes for electrons! 🏠Each shell is a specific layer that can hold a limited number of electrons, kind of like an apartment building! The closest shell to the nucleus can hold 2 electrons. The next one can hold 8, the next 18, and so on! Each shell has a unique level of energy. When electrons move between these shells, they can give off or absorb energy, which is really important in chemistry and physics! 🌟Each shell helps define how an atom interacts with others.
Electron Shells In Periodic Table
The periodic table is a special chart that lists all known elements! 🌍The position of an element can tell us about its electron shells. Elements in the same column share similar properties because they have the same number of electrons in their outermost shell! For example, all the elements in Group 1 (like sodium, Na) have 1 electron in their outer shell. This makes them very reactive! 💥Isn't it cool how shells help scientists understand the different elements?
Comparison With Other Atomic Models
There are different models of atoms, just like different ways of drawing pictures! 🎨One famous model is the Bohr model, which shows electrons in circular orbits around the nucleus. However, the electron shell model is more accurate because it considers the probability of where electrons might be found. 🌌Unlike the hard paths of the Bohr model, the shell model allows for more complexity, showing how electrons can live in areas of space rather than definite paths. These different models help scientists understand the tiny world of atoms! 🌠
Interactions Between Electron Shells
Electrons don’t just hang out in their shells; they interact with each other! 🌐When atoms come close together, their electron shells can overlap, allowing electrons to be shared or transferred. This is how bonds form, creating molecules like water (H₂O) and carbon dioxide (CO₂). ☁️ The way electrons interact can affect everything from how strong a bond is to whether a substance is a solid, liquid, or gas. Such interactions are key in solving puzzles in chemistry!
Applications Of Electron Shell Theory
Electrons and their shells aren’t just fun to learn about; they are also very useful! 🛠️ Scientists use electron shell theory in fields like medicine, electronics, and even environmental science. For instance, understanding how electrons work helps create special materials for batteries, making tech gadgets possible. 📱Electron shell theory also helps in creating medicines by showing how different atoms will react with our bodies, making it essential for doctors and scientists!
Importance Of Electron Shells In Chemical Properties
Electron shells are super important for understanding how elements behave in chemistry! 💡The outermost shell is called the valence shell, and it has the electrons responsible for chemical reactions. Atoms like to have their shells full, so they often bond with other atoms to share or transfer electrons! When they do this, they form different substances. For example, sodium (Na) and chlorine (Cl) bond together to make table salt (NaCl)! 🧂Understanding these shells helps explain why certain materials are reactive and others are not.
Electron Shell Quiz
Learn more about Electron Shell



