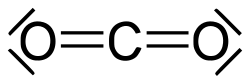Carbonyl Facts For Kids
A carbonyl group is a functional group in organic chemistry that consists of one carbon atom double-bonded to one oxygen atom (C=O), influencing the properties and reactions of many organic compounds.


Do more with AI
Introduction
Welcome to the world of carbonyl groups! 🌍Carbonyl groups are a special pair of atoms that are super important in chemistry. They consist of a carbon atom (C) that is double-bonded to an oxygen atom (O), making a "C=O" shape. You can think of carbonyls as the "C" and "O" buddies! 🤝These groups are found in many different compounds that are used in everyday life. Learning about them helps us understand everything from smells to medicines! So, let’s dive in and explore what carbonyls are and why they are so cool! 🌟
Images of Carbonyl
Carbonyl Group In Nature
Did you know that carbonyl groups are all around us in nature? 🌻Plants and animals contain carbonyl compounds! For instance, the smell of ripe fruits comes from carbonyls like aldehydes, which attract animals to eat them! 🍏In addition, many essential oils derived from plants contain these compounds. Honey contains carbonyls too, which is why it has its sweet taste! 🍯It's fascinating how these small groups play a big role in our environment and how living things interact with one another!
What Are Carbonyl Groups?
A carbonyl group is a functional group found in many molecules! Its formula is C=O, which means a carbon atom is double-bonded to an oxygen atom. This arrangement gives carbonyls special properties. 🌈They can be found in things like sugars, acids, and drugs! The bond between carbon and oxygen is strong, which is why carbonyl groups play important roles in chemical reactions. They can be compared to a doorway that helps molecules enter different phases! 🚪Understanding carbonyl groups can help us learn about how different substances interact with each other!
Types Of Carbonyl Compounds
Carbonyl compounds come in various shapes and sizes! The main types are aldehydes and ketones. 🥇Aldehydes have the carbonyl group at the end of the molecule, like in formaldehyde, found in some household products. 🌿Ketones, on the other hand, have the carbonyl group in the middle, like in acetone, found in nail polish remover! 💅There's also carboxylic acids, which have a carbonyl group and a hydroxyl group (-OH) together! 🤯Each type has unique properties and uses, making them special in the world of chemistry!
Detection And Analysis Of Carbonyls
Scientists need to detect carbonyls in various substances, and they have special techniques for this! 🔬One common method is using a test called the "2,4-Dinitrophenylhydrazine" (or DNPH) test. In this test, when carbonyls are present, they create a colorful compound! 🌈Another technique is mass spectrometry, which helps scientists figure out the exact type of carbonyl compounds in a sample. 🧪Detecting and analyzing these groups is crucial for understanding environmental impacts and human health!
Reactions Involving Carbonyl Groups
Carbonyl groups are like superstars in chemical reactions! ⭐They can easily participate in reactions that change their structure and create new compounds. One common reaction is called "nucleophilic addition," where other molecules come in and add to the carbonyl. For example, they can form alcohols! 🥂Another cool reaction is oxidation, which can turn an aldehyde into a carboxylic acid! Remember, when carbonyls react, they can change into something entirely new, helping create different substances we see every day! 🔄
Role Of Carbonyls In Biological Systems
Carbonyl groups are super important for living organisms! 🌱They appear in sugars, which are necessary for energy. 🥕For instance, glucose, a type of sugar, has a carbonyl group, helping our bodies get the energy we need to play! ⚽Additionally, hormones like testosterone and estrogen also contain carbonyl groups, playing vital roles in growth and development! 🍀Our bodies rely on carbonyls to function well, making them essential for life!
Historical Discoveries Related To Carbonyls
The fascinating world of carbonyls has a rich history! 🔍In 1770, a scientist named Johann Wolfgang Döbereiner discovered the first carbonyl compound: acetaldehyde, found in ripening fruits! 🍎Later, in 1833, French chemist Anselme Payen isolated glucose, realizing its importance in energy! 💡Throughout history, scientists have been discovering more about carbonyls and how they impact chemistry and biology! Every tiny molecule has a big story to tell!
Structural Characteristics Of Carbonyl Groups
Carbonyl groups are distinct due to their unique structure. ✨The carbon atom in a carbonyl group is sp² hybridized, which means it forms three bonds: two with neighboring atoms and one double bond with the oxygen (C=O). 🧬The oxygen atom has a lone pair of electrons, making it a good reaction partner! Most carbonyl groups also create a flat, planar shape, which helps other molecules interact with them easily. The structure is like a platform for chemical reactions, making it special in the chemistry world! 🎡
Applications Of Carbonyl Compounds In Industry
Carbonyl compounds have many important uses in industry! 🏭For example, acetone (a ketone) is used as a solvent in nail polish remover and paint! 🎨Additionally, carbonyls are used to make fragrances for perfumes. 🌹The sweet smell comes from various carbonyl fragrances! Some carbonyl compounds are also key ingredients in making medicines, helping us stay healthy! 💊With their versatility, carbonyls play an essential role in many products we use every day!
Carbonyl Quiz
Learn more about Carbonyl