Enthalpy Facts For Kids
Enthalpy is the total energy in a system, combining internal energy and the energy needed for pressure and volume.


Do more with AI
Introduction
Enthalpy is a super cool topic in science that helps us understand how energy works! 🔋It is the sum of a system's internal energy (the energy inside something) and the energy from its pressure and volume. Think of a balloon! When you blow it up, you add energy. 🎈Enthalpy can help scientists study heat in chemical reactions, like baking cookies! 🍪By measuring enthalpy, they can understand how much energy is used or released. Enthalpy is measured in a unit called joules (J) and it plays a big role in chemistry and physics! 🌍
Gallery of Enthalpy
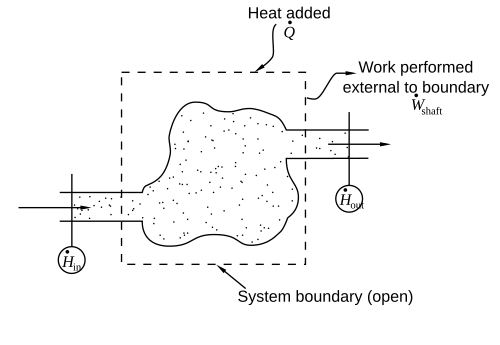
Enthalpy Diagrams
Enthalpy diagrams are like maps for understanding energy changes in reactions! 🗺️ These diagrams usually show the enthalpy of the reactants and products during a chemical reaction. The height of the lines on the diagram shows how much energy is present. If the products are higher than the reactants, it's an endothermic reaction (absorbs energy) 🔼. If the products are lower, it's an exothermic reaction (releases energy) 🔽. These diagrams help scientists visualize energy changes easily, making chemistry even more engaging! ⚗️
Units Of Enthalpy
Enthalpy is measured in joules (J), which is a unit for energy. ⚡Imagine joules as little energy points! The more joules something has, the more energy it has! For bigger amounts, we can use kilojoules (kJ), which is equal to 1,000 joules. So, when scientists talk about heat produced in a reaction, they might say "100 kJ." That means there are 100,000 joules of energy! 🎊It helps them understand how much energy is being used or produced in different processes.
The Enthalpy Formula
To calculate enthalpy, we use a special formula! 🤓It looks like this: H = U + PV. Here, "H" is enthalpy, "U" is the internal energy, "P" is the pressure, and "V" is the volume. Let's break it down! 😃Internal energy is all the energy inside a substance, while pressure is how hard the particles push against the walls of their container and volume is how much space the particles take up. By adding these together, we find out how much energy is in our "energy bag"! 🎉
Definition Of Enthalpy
Enthalpy can be thought of like a big energy bag! 🎒It helps scientists figure out how much energy is stored in something, and how that energy changes when things happen, like boiling water or mixing ingredients. 🔥It's like when you heat soup; the soup's energy changes, but it also takes into account how much is in the pot. The term "enthalpy" comes from the Greek word "enthalpos," meaning "to put heat into." So, it's all about heat and energy! 🥵
Enthalpy And Thermodynamics
Enthalpy is an important part of thermodynamics, which is the study of energy and heat flow! 🌈It helps us understand how energy moves and changes in different systems. In thermodynamics, we often talk about three "laws" that explain energy behavior. The first law says energy cannot be created or destroyed, only changed! 🔄Enthalpy is used to help explain these changes. By learning thermodynamics, we can understand everything from engines to weather patterns. How cool is that? 🌌
Enthalpy In Chemical Reactions
In chemical reactions, enthalpy helps us see how energy is used or released! 🌟We can have two types: exothermic reactions, which give off heat, and endothermic reactions, which absorb heat. For example, when you light a match, it gives off heat (exothermic) 🔥. But did you know that making ice cream requires ice and salt to absorb heat energy (endothermic)? 🍦By studying these changes, scientists learn how energy flows in different reactions, making chemistry even more exciting! 🎈
Measurement Of Enthalpy Changes
Scientists use a special tool called a calorimeter to measure enthalpy changes! 🌡️ This tool helps them see how much energy is added or released during a reaction. Imagine putting your hot chocolate in a special cup that measures heat! ☕When you stir in marshmallows, some heat is released as they melt, and the calorimeter helps calculate how much! Knowing these changes is super helpful for understanding reactions, like how much energy is needed to cook food or cool it down! ❄️
Applications Of Enthalpy In Real Life
Enthalpy is everywhere in our daily lives! 🌍It is used in cooking, heating homes, and even weather forecasting! 🍲When cooking, chefs use enthalpy to know how much energy is needed to bake, boil, or fry food! 🔥Engineers use enthalpy to design heating and air-conditioning systems to keep us comfortable at home. Plus, meteorologists use it to check how heat affects the weather! ☀️ So, the next time you enjoy warm bread or stay cozy in your house, remember that enthalpy plays a big role in all that magic! 🎉
Comparative Analysis Of Enthalpy And Other Thermodynamic Quantities
Enthalpy is just one of many important concepts in thermodynamics! 🌈Other quantities include internal energy (which is all the energy inside) and entropy (which tells us about disorder in a system). Enthalpy combines internal energy and factors like pressure and volume, so it's perfect for measuring heat in reactions. 🔥While internal energy focuses only on the energy inside, enthalpy also accounts for how that energy interacts with the surroundings. Understanding all these concepts helps scientists better understand energy and its many forms in the world! 🌍✨
![T − s diagram of nitrogen.[21] The red curve at the left is the melting curve. The red dome represents the two-phase region with the low-entropy side the saturated liquid and the high-entropy side the saturated gas. The black curves give the T − s relation along isobars. The pressures are indicated in bar. The blue curves are isenthalps (curves of constant enthalpy). The values are indicated in blue in .mw-parser-output .sfrac{white-space:nowrap}.mw-parser-output .sfrac.tion,.mw-parser-output .sfrac .tion{display:inline-block;vertical-align:-0.5em;font-size:85%;text-align:center}.mw-parser-output .sfrac .num{display:block;line-height:1em;margin:0.0em 0.1em;border-bottom:1px solid}.mw-parser-output .sfrac .den{display:block;line-height:1em;margin:0.1em 0.1em}.mw-parser-output .sr-only{border:0;clip:rect(0,0,0,0);clip-path:polygon(0px 0px,0px 0px,0px 0px);height:1px;margin:-1px;overflow:hidden;padding:0;position:absolute;width:1px} kJ /kg. The specific points a, b, etc., are treated in the main text.](https://upload.wikimedia.org/wikipedia/commons/thumb/0/03/Ts_diagram_of_N2_02.jpg/500px-Ts_diagram_of_N2_02.jpg)



