Chemical Reaction Facts For Kids
A chemical reaction is a process that transforms one set of chemical substances (reactants) into another set (products).
Do more with AI
Introduction
A chemical reaction is like a magic show where one thing turns into another! 🎩✨ This happens when chemicals, like baking soda or vinegar, mix together and create something new. For example, when a candle burns, the wax changes into smoke and gases. These changes can sometimes make things bubble, change color, or even produce heat! 🔥Chemical reactions happen all around you, from cooking food 🍳 to blowing up a balloon with a fizzy drink! 🥤Let's explore the amazing world of chemical reactions and discover how they work!
Gallery of Chemical Reaction
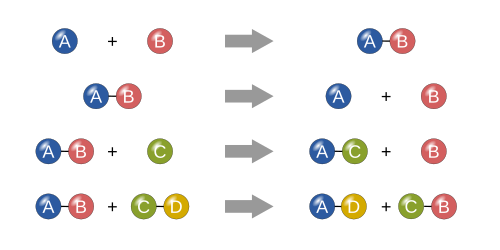
Reaction Mechanisms
A reaction mechanism describes the step-by-step process of how a chemical reaction happens! 🛠️ Imagine following a recipe; you mix ingredients in a specific order to make something delicious. 🍰In chemistry, certain steps happen at different times and can involve tiny particles called intermediates. These can change how fast or slow the reaction occurs. Scientists study reaction mechanisms to understand better how to make desired products, like creating special medicines or fuels! 🚀Knowing the steps helps us improve reactions and invent amazing new things!
Chemical Reaction Kinetics
Chemical reaction kinetics is a fancy way of talking about how fast or slow a reaction happens! 🕒Some reactions happen quickly, like fizzing soda, while others take a lot longer—like rust forming on a bike. 🛴Factors that affect speed include temperature, concentration (how much stuff is mixed), and surface area (how much of a substance is exposed). Higher temperatures usually make reactions happen faster, like when you cook soup! 🍲Scientists use kinetics to predict and control how reactions occur, making them crucial for many experiments!
Types Of Chemical Reactions
There are several types of chemical reactions, and they all do different things. Here are four fun ones:
1. Synthesis: Two or more things join together, like making water from hydrogen and oxygen! 🌊
2. Decomposition: One thing breaks down into smaller parts, like when a banana ripens and gets mushy! 🍌
3. Single Replacement: One element takes the place of another in a compound, like a friend swapping toys! 🔄
4. Double Replacement: Two compounds exchange parts, like trading stickers with friends! 🌟Each reaction is special and teaches us how matter changes in exciting ways!
Balancing Chemical Equations
When scientists write chemical equations, they need to balance them! ⚖️ This means having the same number of atoms on both sides of the equation. For example, in the reaction to make water (H₂ + O₂ → H₂O), we need two hydrogen atoms for every one oxygen atom. This keeps the Law of Conservation of Mass in check! 🧪Balancing ensures that nothing is lost or gained in the reaction. Learning to balance equations is an essential part of chemistry and is like solving a fun puzzle! 🧩
The Law Of Conservation Of Mass
Did you know that in a chemical reaction, mass is never lost? This is called the Law of Conservation of Mass! ⚖️ It means the weight of the reactants (the things we mix) equals the weight of the products (the new things we make). For example, if you have 10 grams of baking soda and you mix it with vinegar, you will get bubbles and foam, but the total weight will still be 10 grams! 🥳This law helps scientists make sure they account for everything during experiments and ensures that nothing just disappears! 💡
Factors Affecting Reaction Rates
Many factors can change how fast a chemical reaction happens! Here are some important ones: 🔍
1. Temperature: Higher temperatures speed up reactions, as particles move faster! 🌡️
2. Concentration: More reactants make reactions happen quicker—think of crowded dance parties! 💃
3. Surface Area: Smaller pieces have more space to react, just like cutting up fruit! 🍓
4. Catalysts: Helpers, called catalysts, can speed up reactions without changing themselves. 🔧Understanding these factors helps scientists create faster reactions to make new products efficiently!
Applications Of Chemical Reactions
Chemical reactions are all around us and play major roles in our daily lives! 🌍Here are some fun applications:
1. Cooking: When we bake a cake, different ingredients react to make it rise and taste delicious. 🥮
2. Medicine: Doctors use chemical reactions to create medicines that help fight illness. 💊
3. Cleaning: Soap and detergents work through chemical reactions to remove dirt and grease. 🧼
4. Energy: Cars and power plants use chemical reactions to produce energy that keeps everything moving. ⚡Exploring these applications shows how chemistry helps make our world a better place!
Exothermic And Endothermic Reactions
Chemical reactions can be warm or cool, just like a hot cup of cocoa or ice cream! ☕🍦 When a reaction gives off heat, it’s called *exothermic*. For example, when you light a campfire, it gets hot as wood burns! 🔥On the other hand, *endothermic* reactions absorb heat, making things feel cooler. 🌬️ An example is when you mix baking soda and vinegar, the solution gets cold as it fizzes! Understanding these types helps scientists create amazing things, like instant cold packs for injuries! ❄️
Real-world Examples Of Chemical Reactions
Chemical reactions are happening around us every day! 🌞Here are some cool examples:
1. Baking: When baking bread, yeast ferments, releasing carbon dioxide—which makes the bread rise! 🍞
2. Rusting: Iron reacts with air and water to form rust, which can damage metal. 🌧️
3. Photosynthesis: Plants use sunlight and carbon dioxide to create food and oxygen. 🌱
4. Fireworks: Chemical reactions between specific compounds produce colorful explosions in the sky! 🎆These examples show how chemistry is a part of our environment in fascinating ways!
Safety Considerations In Chemical Reactions
Safety is super important when doing chemical reactions! 🦺Always wear goggles and gloves to protect yourself. Here are some more tips:
1. Supervision: Always have an adult around when you are mixing chemicals or doing experiments! 👩🔬
2. Ventilation: Make sure the area is well-ventilated to avoid harmful fumes. 🌬️
3. Read Labels: Always check labels for warnings and instructions on chemicals. ⚠️
4. Clean Up: After experiments, clean up spills and dispose of materials safely! 🧹Following these safety tips helps keep everyone safe while exploring the exciting world of chemistry!