Radioactive Decay Facts For Kids
Radioactive decay is the process by which unstable atomic nuclei lose energy by emitting radiation, leading to the transformation of one element into another over time.
Do more with AI
Introduction
Radioactive decay is a fascinating process where unstable atoms change into more stable ones! 🌍Atoms are the tiny building blocks of everything around us. Some atoms, like Uranium-238 and Radon-222, are unstable, meaning they can break down or "decay" over time. When they do, they release energy and particles. This can be helpful for things like energy and medicine. In simple terms, think of it like a Lego tower that slowly comes apart to build something new! 🏗️ Radioactive decay is found everywhere on Earth, and it’s a topic scientists study to learn more about our world.
Gallery of Radioactive Decay
The Decay Process
The decay process is like a clock that never stops! 🕰️ When an unstable atom starts to decay, it shrinks a little and releases particles or energy! Each type of decay happens differently, but they all share a common goal: to reach a stable state. Think of a balloon that keeps losing air until it finally becomes limp! 🌬️ Over time, radioactive materials can transform into something completely different. Scientists have special detectors that can measure the decay and see how long it takes for this to happen. This helps them learn about how atoms work and what happens to them as they change.
Radiometric Dating
Radiometric dating is an exciting technique scientists use to figure out the ages of rocks, fossils, and even Earth itself! 🌎By measuring the amounts of radioactive isotopes and their stable products, scientists can calculate how many years have passed since the rock was formed. For example, using Potassium-40 that decays into Argon-40, they can determine the age of volcanic rocks. ⛰️ This method has helped scientists discover that Earth is about 4.5 billion years old! Radiometric dating gives us a timeline of Earth’s history, helping us understand how our planet has changed over millions of years! 🕰️
Types Of Radioactive Decay
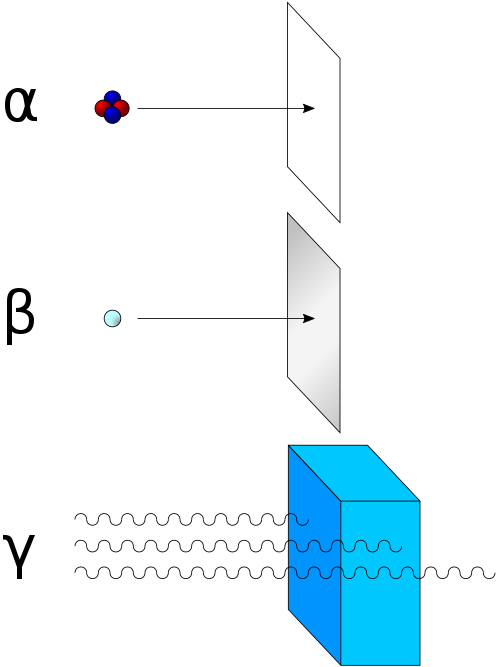
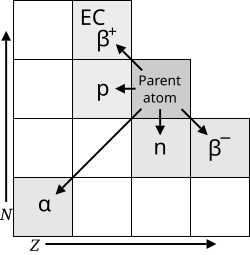
There are three main types of radioactive decay: alpha decay, beta decay, and gamma decay! 😮In alpha decay, an atom releases two protons and two neutrons, making the atom smaller. For example, Uranium-238 decays into Thorium-234. In beta decay, a neutron changes into a proton and releases an electron, turning the atom into a new element. An example of this is Carbon-14 turning into Nitrogen-14. Finally, gamma decay releases energy in the form of gamma rays, which are very powerful! 🌌These different types of decay show that atoms can change in exciting ways!
Half-life And Its Applications
Half-life is a special term that helps us understand how long it takes for half of the radioactive atoms in a sample to decay! ⏳For example, if you have a pile of 100 atoms of Carbon-14, after 5,730 years, only 50 atoms will remain unchanged! Scientists use this concept to help date ancient rocks and fossils. 🌋The half-life can vary greatly: Uranium-238 has a half-life of about 4.5 billion years! Learning about half-lives helps us understand how old things are and gives important clues in science and medicine, like how long a radioactive medicine stays in the body!
Uses Of Radioactive Decay In Medicine
Radioactive decay has many important uses in medicine! 🏥Doctors use radioactive materials to help diagnose and treat illnesses. For example, a form of iodine called Iodine-131 can be used to treat thyroid problems! 🌟When it decays, it releases energy that helps destroy the unhealthy thyroid cells. Another method is PET scans! These scans use a tiny amount of a radioactive substance to look inside our bodies and see how organs are working. 🎈This helps doctors find problems like cancer. Because of radioactive decay, medicine has advanced, making treatments more effective for sick people!
Experimental Methods For Studying Decay
Scientists use many cool methods to study radioactive decay! 🔬One common method is a Geiger counter, which detects radiation levels in the surrounding area. This helps researchers measure how much radiation is being released from a sample! Another exciting way is through nuclear decay chains—when one radioactive element decays into another, scientists can follow this chain to learn more about the process. 🔄They also use advanced technology like particle accelerators to create new isotopes for testing! By experimenting with radioactive decay, scientists discover new information and enhance our understanding of the atom's life cycle! 🧪
Historical Discoveries In Radioactivity
The study of radioactivity has a rich history! 🌟Marie Curie was a famous scientist who discovered the elements Polonium and Radium! Her work laid the foundation for modern research in radioactivity. In 1896, Henri Becquerel accidentally discovered radioactivity while studying uranium. That discovery opened the door to a whole new field of science! Another major milestone was the discovery of nuclear fission in 1938 by Lise Meitner and Otto Hahn, leading to advancements in energy and medicine. 🌌These incredible scientists helped us understand radioactive decay and its potential uses, shaping the world we live in today!
Safety And Hazards Of Radioactive Materials
While radioactive materials have helpful uses, they can also be dangerous! ⚠️ If people are exposed to high levels of radiation, it can cause harm to their bodies. This is why special safety measures are in place! Places that use radioactive materials must follow strict rules, like wearing protective clothing and staying at a safe distance. 🦺Even small amounts can be harmful, so scientists take these materials very seriously. It's important for everyone to understand the risks, just like wearing a helmet when riding a bike! Safety first ensures that we can enjoy the benefits of radioactive decay without getting hurt!
Radioactive Decay Quiz
Learn more about Radioactive Decay