Triple Point Facts For Kids
The triple point is a special condition in thermodynamics where a substance can exist in solid, liquid, and gas phases at the same time and under specific temperature and pressure conditions.

Do more with AI
Introduction
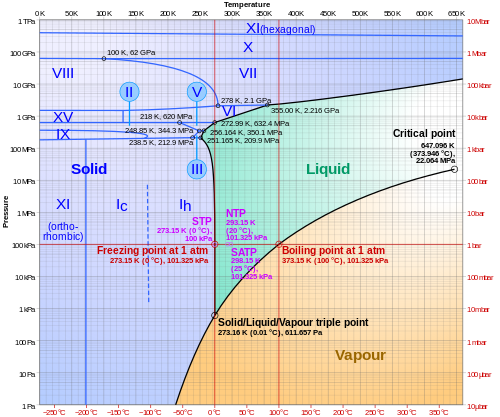
Have you ever wondered how water can be ice, liquid, and steam all at once? 🤔This magical moment is called the "triple point!" A substance's triple point occurs at a specific temperature and pressure where all three phases—solid (ice), liquid (water), and gas (steam)—exist together in harmony. For water, this happens at 0.01°C (32.018°F) and 611.657 pascals of pressure. 🥶💧💨 The triple point helps scientists understand how substances behave under different conditions. Let's dive deeper into this fascinating concept!
Gallery of Triple Point
Common Misconceptions
Many people think that only ice, water, and steam can be in a triple point. ❌But that's not true! Other substances, such as carbon dioxide and sulfur, also have triple points with different temperatures and pressures. Also, some might believe that all three phases are equally visible and stable at the triple point, but that's not always the case. 🔄In some situations, one phase may be more visible than the others, so not everything is always apparent. Understanding these details helps clarify the amazing science behind phases of matter!
Phases Of Matter Involved
In our everyday lives, we usually see three main phases of matter: solid, liquid, and gas. 🔢Ice represents the solid phase, while water is the liquid phase, and steam is the gas phase. The triple point is where all three can exist together! For water, at the triple point, tiny ice crystals float in water, and steam can be seen all around. 🌬️ By studying these phases, scientists learn how materials transition between different states and how to control those changes. It’s fascinating how one point on a graph can show us so much!
Definition Of Triple Point
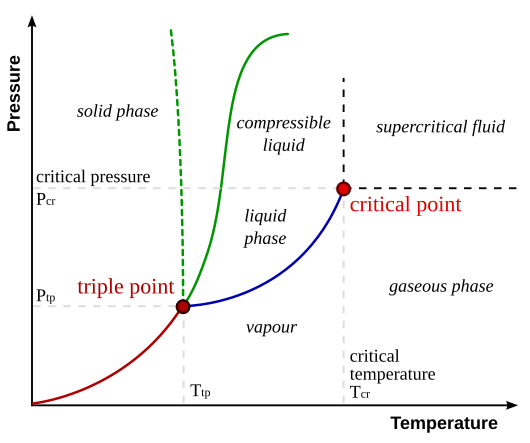
The triple point is a special spot where three phases of a substance come together. 🌟Imagine seeing ice, water, and steam in the same jar! This happens at a precise temperature and pressure. For example, the triple point for water is 0.01°C and 611.657 Pascals. Scientists use this unique spot to study how substances change from solid to liquid and then to gas. 🔄It’s vital for understanding how materials work under various conditions and can help us in many ways!
Experimental Determination
To find the triple point, scientists perform experiments where they carefully control temperature and pressure. 🧪They use special equipment to measure these conditions accurately. For instance, they may put water in a sealed container, then cool it down slowly while adjusting pressure. ❄️ When they achieve the right temperature and pressure, they observe ice, liquid water, and steam together! Experiments like these provide essential data, making it easier for scientists to understand the properties and behaviors of different substances.
Further Reading And Resources
If you're curious to learn more about the triple point and phases of matter, here are some fun resources! 📚Check out books like "Phases of Matter" by Oliver S. and "The Science of Cooking" by Peter L. You can also explore educational websites like NASA's "Phases of Matter" page or read fun articles on National Geographic! 🌍Joining a local science club or visiting a science museum can also offer exciting hands-on experiences and experiments. Keep exploring the wonders of science! 🧪✨
Significance In Thermodynamics
Thermodynamics is all about energy and heat. 🔥The triple point is significant because it helps researchers study how substances react to changes in temperature and pressure. When scientists know a substance's triple point, they can create accurate phase diagrams, which show how a material transitions from solid to liquid to gas. 📊This knowledge is important in various fields like engineering and physical science, helping us understand energy systems, climate, and even cooking! 🍳It helps us find the balance between heat and cold in different materials.
Relationship With Phase Diagrams
Phase diagrams are like maps for different states of matter! 🗺️ They help scientists understand how substances change from solid to liquid to gas as temperature and pressure change. The triple point is a specific point on these diagrams, where all three phases coexist. 🌌In water's phase diagram, for example, the triple point is a key marker showing where ice, water, and steam can be found together. By studying these diagrams, scientists can predict how materials will behave under different conditions!
Historical Context And Discoveries
The concept of the triple point was discovered in the 19th century! 🕒The first scientist to explore it was the French physicist, Pierre-Simon Laplace, who studied how ice and steam could co-exist. Later, in 1860, the German physicist, J. W. Gibbs, expanded the idea, allowing scientists to create accurate phase diagrams. ⚗️ This work helped establish the fundamentals of thermodynamics! Since then, many scientists have used the triple point to explore and discover new materials, finding applications to improve our lives and understanding of the world!
Applications In Science And Industry
The concept of the triple point has many useful applications! 🔌For example, it helps researchers create highly precise thermometers by using substances with known triple points to calibrate measurements. Scientists studying space also use the triple point to understand how materials behave in extreme conditions! 🚀Additionally, manufacturers can control the different phases of materials, like melting or freezing processes, to create better products, such as ice cream or metals! 🍦Whether in a lab or factory, the triple point plays an important role!
Triple Point In Different Substances
Just like water, other substances also have triple points! 🌈For example, the triple point of carbon dioxide (like the fizz in soda) is -56.6°C and 518 kPa. Another cool example is sulfur, which has a triple point of 113°C and 0.106 MPa. Each substance has its own unique triple point, helping scientists learn more about their characteristics. 🤓Studying these different triple points can also help create new materials and improve technology like cooling systems and food preservation!