Transition Metal Facts For Kids
Transition metals are elements in the d-block of the periodic table, known for their unique properties, versatility, and ability to form colorful compounds.
Do more with AI
Introduction
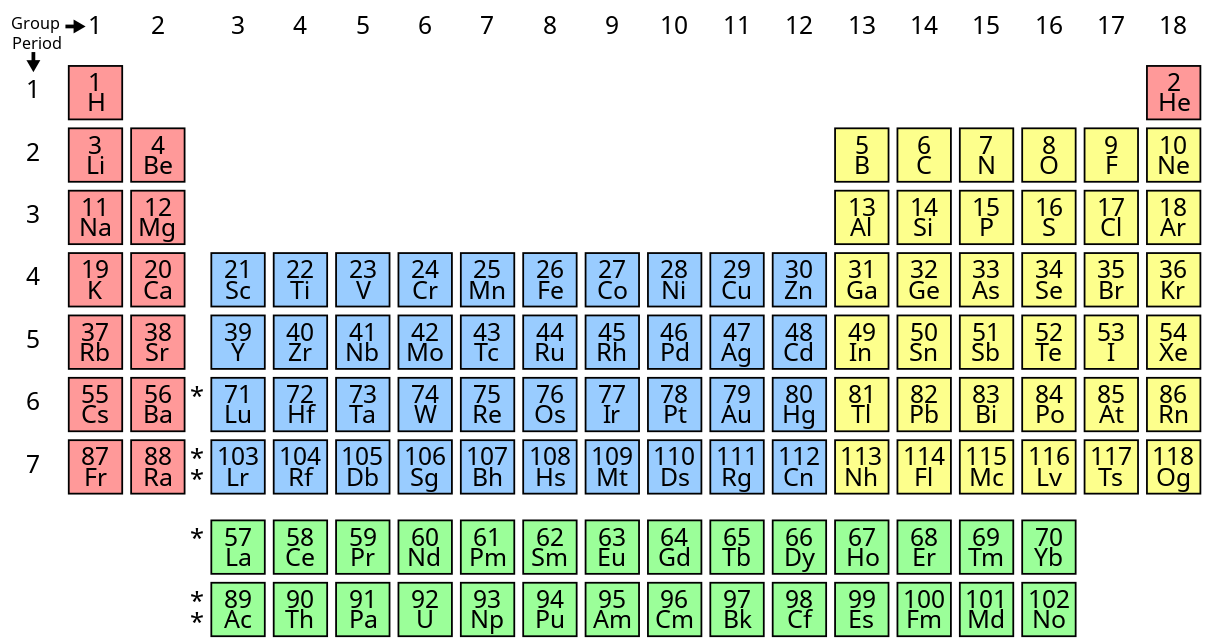
Transition metals are a special group of elements found in the d-block of the periodic table! 🌟These metals include powerful elements like iron (Fe), copper (Cu), and gold (Au). They are located in the middle part of the periodic table, specifically from group 3 to group 12. These metals are unique because they can form colorful compounds and can easily change their electrons. Transition metals are used in many everyday items, like jewelry, coins, and even in cars! 🚗These strong metals help make our world a better place through their amazing properties.
Gallery of Transition Metal
Reactivity And Catalysis
Transition metals are known for their reactivity! ⚡They often participate in chemical reactions, which can help change other substances. Many transition metals act as catalysts, speeding up reactions without getting used up! For example, in cars, platinum is used in catalytic converters to reduce harmful gases. This keeps our air cleaner! 🌍Iron is reactive, too; it rusts when exposed to water and air. Learning about how these metals react not only helps us understand chemistry better but also shows how to use them to solve real-world problems! 🔬
Interesting Facts And Trivia
Here are some fun facts about transition metals! 🎉Did you know that gold, a transition metal, doesn’t rust? It stays shiny forever! 🌟Transition metals can create stunning colors in fireworks; different metals produce various colors, like strontium for red and barium for green! 💥Interestingly, mercury (Hg) is the only metal that is liquid at room temperature! It was once used in old thermometers. 📏Lastly, the famous blue color of the Statue of Liberty comes from its copper metal surface that oxidized over time, turning turquoise! These metals truly have a fascinating world! 🏛️
Comparison With Other Elements
Transition metals are unique compared to other elements! While nonmetals like oxygen (O) are gases at room temperature, most transition metals are solid and shiny. ✨Unlike alkali metals, which are very reactive and soft, transition metals are more stable and have high melting points! They can form colorful compounds, setting them apart from other groups. 🔴🔵 Some other elements don’t have the ability to change oxidation states like transition metals can. This special behavior allows them to play various roles in nature and technology, helping make them one of the coolest groups on the periodic table! 🌟
Biological Roles And Importance
Did you know transition metals are super important for living things? 🦠Elements like iron are found in our bodies and are vital for making hemoglobin, which transports oxygen in our blood! 🌬️ Zinc is another transition metal, essential for our immune system. Copper plays a role in making energy and keeping our bones strong. These metals help us stay healthy and are found in the food we eat, like nuts and beans. So next time you enjoy a healthy meal, remember that transition metals are helping your body function! 🍽️
Applications In Industry And Technology
Transition metals are superstars in industry and technology! 💪They help in creating strong materials, like steel made from iron and carbon. These sturdy materials are used in buildings, cars, and even bridges! Transition metals like titanium are light yet durable, perfect for airplanes. ✈️ In the world of electronics, copper is crucial for wiring and circuit boards. They also help in making batteries, tools, and even catalysts to speed up chemical reactions! Transition metals are everywhere, making our technology work efficiently and safely, and helping us build better products! 🛠️
Environmental Impact And Sustainability
Transition metals play a significant role in our environment! 🌳While they can help create clean energy, like in solar panels with silver (Ag), some metals can also harm nature. Heavy metals like lead (Pb) and mercury (Hg) can pollute water and soil, making it essential to use them wisely. 🌊Scientists are working hard to find sustainable ways to use transition metals, recycling them from older products. This helps protect our planet while still benefiting from their amazing properties. Keeping our environment safe ensures a bright future for all living creatures! 🐾
General Properties Of Transition Metals
Transition metals have some cool properties! 🎉They usually have high melting and boiling points, meaning they can stay solid in really hot places. They are also excellent conductors of electricity and heat, which is why they are often used in wires and appliances. Most transition metals are shiny and can be easily shaped into different forms, like gold jewelry! ✨They can form different colors when mixed with other substances, making them super fun for artists using paint and dyes. Their diverse behaviors help them play important roles in various industries!
Common Compounds And Coordination Chemistry
Transition metals love to bond with other elements, forming cool compounds! ⚗️ One example is salt, created with sodium (Na) and chlorine (Cl). Transition metals like copper can form compounds like copper sulfate, which is used for blue fertilizers! 🌿Coordination chemistry is all about how metals bond with other molecules like water or ammonia. This helps transition metals to create complex structures. For instance, hemoglobin, found in our blood, contains iron and helps carry oxygen! 🩸The study of these compounds shows how essential transition metals are in chemistry and biology.
Electron Configuration And Oxidation States
Transition metals are unique because they can have different electron configurations. 🔍Each transition metal has a specific number of electrons in their outer shells. This means they can lose and gain electrons easily! This process is called oxidation states. For example, iron can be in the +2 or +3 state, while copper can be +1 or +2. These different states allow transition metals to form various compounds and make them crucial in chemical reactions. Learning about their electron configurations helps us understand why they behave the way they do in nature! 🌈
Historical Discovery And Importance In Chemistry
Transition metals have a rich history in chemistry! 🧪The first transition element, iron, has been used for over 3,000 years to make tools and weapons. Copper was one of the first metals humans learned to use, shaping the Copper Age around 5,000 BC. 📅Many scientists, like Dmitri Mendeleev, studied these metals to create the periodic table we use today. Understanding transition metals has allowed us to develop countless technologies and scientific advancements. Their discovery marked the beginning of many exciting developments in chemistry, shaping our modern world! 🌎
Transition Metal Quiz
Learn more about Transition Metal