Hydroxyl Facts For Kids
In chemistry, a hydroxyl group is a functional group with the formula -OH, composed of one oxygen atom bonded to one hydrogen atom, playing a vital role in various compounds and reactions.


Do more with AI
Introduction
Welcome to the fascinating world of hydroxyl! 🌍A hydroxyl group is a tiny team made of one oxygen atom and one hydrogen atom, linked together like best friends. Its chemical formula is −OH. You can find hydroxyl in many places, like in water (H₂O) and alcohols. Hydroxyl groups are very important in chemistry because they help create many different substances! They’re found in nature, foods, and even in our bodies. Let’s explore how these little buddies work together and why they are so special! 🌟
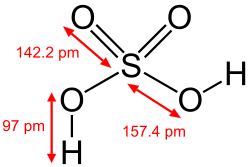
Gallery of Hydroxyl
Hydroxyl In Biochemistry
In biochemistry, hydroxyl groups play important roles in living organisms! 🧬They are found in vital molecules like carbohydrates, proteins, and nucleic acids (like DNA). For example, in sugars, hydroxyl groups help store energy that cells use. 🥪In DNA, they help with the structure, allowing it to carry genetic information. Hydroxyl groups also help enzymes work, speeding up chemical reactions inside our bodies. This is how our body gets energy from food! Learning about hydroxyl groups opens the door to understanding how all living things work, making it a key part of science and life! 🌱🌞
Hydroxyl Group In Alcohols
Hydroxyl groups are the life of the party when it comes to alcohols! 🍹Alcohol is a type of compound that has a hydroxyl group attached to it. For example, ethanol is the alcohol found in drinks like beer and wine. The presence of the hydroxyl group makes alcohols polar, which means they can mix well with water! This is why alcoholic drinks can be watery. Hydroxyl groups give alcohols their unique properties, like the way they taste and how they make you feel. Remember, while some alcohol is fun, it must be consumed carefully and never by kids! 🚫🍸
Hydroxyl Group And Ph Levels
Hydroxyl groups can influence pH levels! 🧪pH is a way to measure how acidic or basic a solution is, ranging from 0 (very acidic) to 14 (very basic). If a solution has more hydroxyl groups, it usually becomes more basic. This happens because hydroxyl groups can help neutralize acids. For example, if you add baking soda, which contains hydroxyl ions, to a solution, it can raise the pH and make it less acidic. Understanding pH is important in chemistry, as it helps scientists and chefs create the right conditions for reactions. So, hydroxyl groups help keep things balanced! ⚖️
Properties Of Hydroxyl Group
Hydroxyl groups have some cool properties! 🌡️ First, they are polar, which means they have a slight electrical charge. This helps them mix with water, making them great for dissolving things. Hydroxyl groups can also form hydrogen bonds, making them sticky! This stickiness helps molecules like sugar dissolve in water. When hydroxyl groups are present in a compound, they can change how that compound behaves. For example, adding a hydroxyl group to a molecule can make it a liquid at room temperature or help it smell nice. Isn't that neat? 🥳
Chemical Structure Of Hydroxyl
The chemical structure of a hydroxyl group is simple but powerful! 💪It is made of one oxygen atom (O) and one hydrogen atom (H), connected by a covalent bond. In this bond, the oxygen shares its electrons with hydrogen, creating a strong relationship between them. This is what gives hydroxyl its -OH label! You can imagine the oxygen as the big brother taking care of the little hydrogen. When they join together, they help make things like sugars, acids, and alcohols. Understanding this structure helps us learn about how other molecules are built! 🔍
Hydroxyl As A Functional Group
Hydroxyl is more than just a cute group; it’s a functional group! 🤩Functional groups are special parts of molecules that define their properties and reactions. Because the hydroxyl group is so common, it can be found in many chemical compounds. Just like how a hat can change your look, adding a hydroxyl group can change how a molecule behaves! 🧢This is why scientists study hydroxyl groups closely. Understanding them helps us create new medicines, materials, and even delicious foods. Hydroxyl groups are like tiny superheroes hiding inside bigger molecules! 🦸♂️
Hydroxyl In Phenolic Compounds
Phenolic compounds are special substances that also contain hydroxyl groups! 🧪They are made up of a benzene ring (a special ring-shaped structure) and one or more hydroxyl groups. You can find phenolic compounds in plants and even in some foods. For example, the delicious chocolate you love contains phenolic compounds, which give it a rich flavor and smell! 🍫These compounds have important roles in protecting plants from pests and helping them stay healthy. Scientists study phenolic compounds to learn more about their effects on our health and the environment! 🌱
Hydroxyl's Impact On Solubility
Hydroxyl groups play a big role in how things dissolve in liquids! 💧They are polar, which means they can help pull other polar molecules (like salt or sugar) into solutions. Imagine trying to mix a spoonful of sugar in your lemonade! 🍋The hydroxyl groups in sugar help it break up and spread out in the water. This makes the sugar easier to dissolve. Hydroxyl groups are like little helpers that make everything mix smoothly. When compounds have hydroxyl groups, they are generally more soluble in water. This is super important for everyday life, like when we drink beverages or cook! 🥤
Natural Occurrences Of Hydroxyl
Hydroxyl groups are everywhere in nature! 🌿You can find them in plants, animals, and even in the air. They are present in most fruits and vegetables, making them tasty and healthy! 🍎For example, apples contain hydroxyl groups in their sugars, which help make them sweet. Hydroxyl groups are also found in water – H₂O has two hydrogen atoms and one oxygen atom! In addition, they help make compounds like phenols, which protect plants from diseases. Understanding where hydroxyl groups occur in nature helps scientists learn about the environment and how everything works together! 🌏
Roles Of Hydroxyl In Organic Chemistry
In the world of organic chemistry, hydroxyl groups play a superstar role! 🌟Organic chemistry studies carbon-based compounds, and hydroxyl groups are found in many of them. They create connections that help build larger molecules, like sugars and alcohols. Hydroxyl groups also help compounds react with one another, making new materials. Think of them like tiny builders constructing a big LEGO set! 🏗️ This is why scientists love studying hydroxyl groups; they help us understand how different substances work and how to create new things from them! 🎉